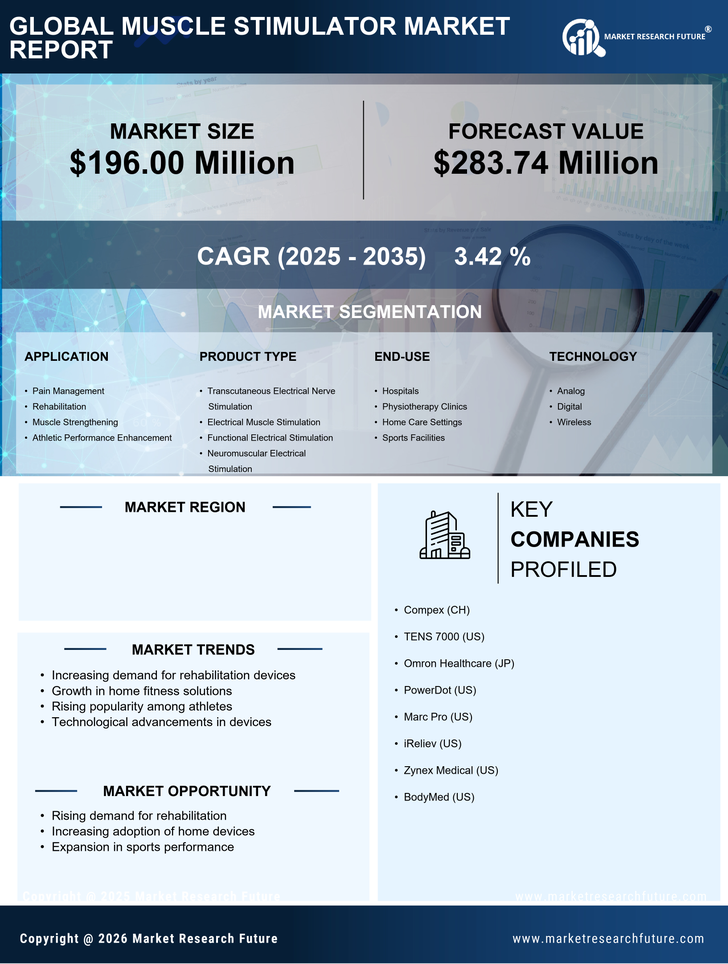
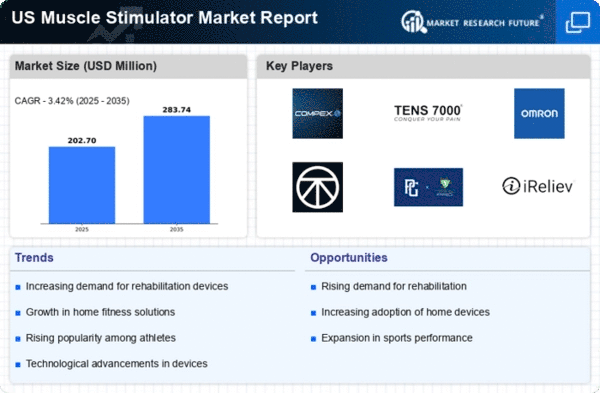
US Healthcare Market Segmentation
Healthcare By Application (USD Million, 2025-2035)
- Pain Management
- Rehabilitation
- Muscle Strengthening
- Athletic Performance Enhancement
Healthcare By Product Type (USD Million, 2025-2035)
- Transcutaneous Electrical Nerve Stimulation
- Electrical Muscle Stimulation
- Functional Electrical Stimulation
- Neuromuscular Electrical Stimulation
Healthcare By End-use (USD Million, 2025-2035)
- Hospitals
- Physiotherapy Clinics
- Home Care Settings
- Sports Facilities
Healthcare By Technology (USD Million, 2025-2035)
- Analog
- Digital
- Wireless