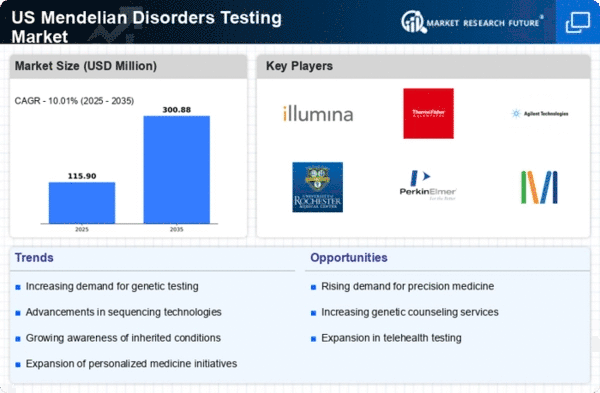
US Chemicals and Materials Market Segmentation
Chemicals and Materials By Type (USD Million, 2025-2035)
- carrier testing
- diagnostic testing
- new-born screening
- predictive & pre-symptomatic testing
- prenatal testing
- others
Chemicals and Materials By Disorder Type (USD Million, 2025-2035)
- cystic fibrosis
- sickle cell anemia
- severe combined immunodeficiency (SCID)
- Tay-Sachs disorder
- polycystic kidney disorder
- Gaucher's disease
- Huntington's disease
- neurofibromatosis
- thalassemia
- familial hypercholesterolemia
- others
Chemicals and Materials By End User (USD Million, 2025-2035)
- hospitals
- ambulatory surgical centers (ASCs)
- specialized clinics