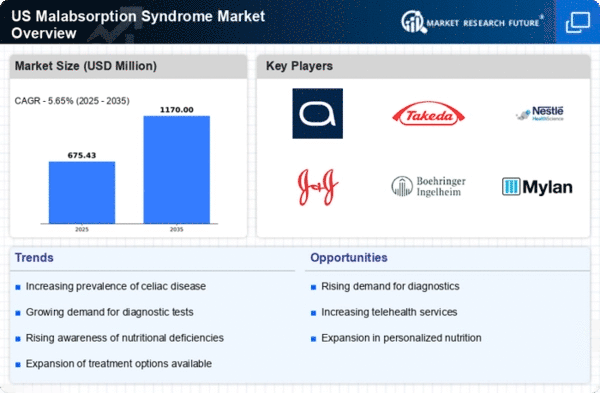
US Medical Device Market Segmentation
Medical Device By causative diseases (USD Million, 2025-2035)
- Cystic fibrosis
- Parasitic diseases
- Intestinal disorders
- Celiac disease
- Others
- Lactose intolerance
- Whipple’s disease
- Tropical sprue
- Others
Medical Device By diagnosis (USD Million, 2025-2035)
- Imaging studies
- CT scan
- Endoscopic retrograde cholangiopancreatogram (ERCP)
- Magnetic resonance cholangiopancreatography (MRCP)
- Abdominal x-ray
- Hematologic tests
- Other tests
- Test of fat malabsorption
- Tests of carbohydrate absorption
- Schilling test
- D-xylose test
- Test of bile salt absorption
- C-d-xylose breath test
Medical Device By treatment (USD Million, 2025-2035)
- Gluten-free diet
- Anti-inflammatory drugs
- Antibiotics
- Nutritional supplements
- Protease and lipase supplements
- Antidiarrheal agents
- Others
Medical Device By end-user (USD Million, 2025-2035)
- Hospitals
- Academic and Research Institutes
- Pharmaceutical companies
- Nutrition Clinics
- Others
.png)