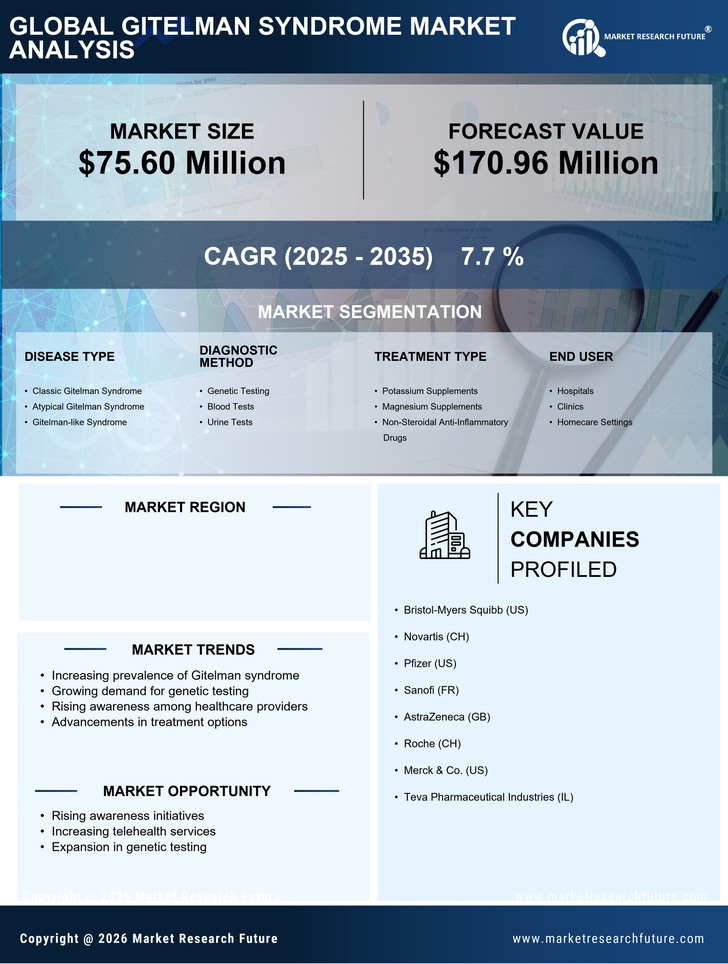
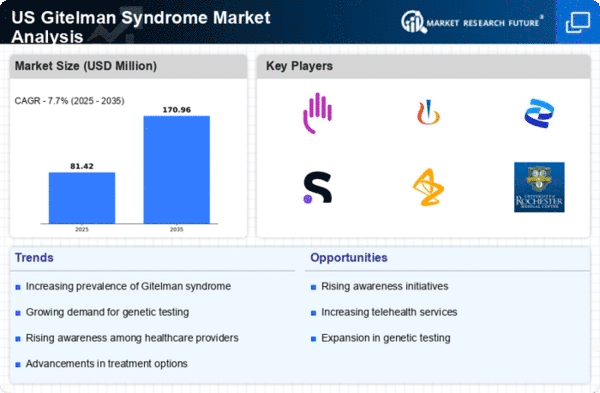
US Chemicals and Materials Market Segmentation
Chemicals and Materials By Disease Type (USD Million, 2025-2035)
- Classic Gitelman Syndrome
- Atypical Gitelman Syndrome
- Gitelman-like Syndrome
Chemicals and Materials By Diagnostic Method (USD Million, 2025-2035)
- Genetic Testing
- Blood Tests
- Urine Tests
Chemicals and Materials By Treatment Type (USD Million, 2025-2035)
- Potassium Supplements
- Magnesium Supplements
- Non-Steroidal Anti-Inflammatory Drugs
Chemicals and Materials By End User (USD Million, 2025-2035)
- Hospitals
- Clinics
- Homecare Settings