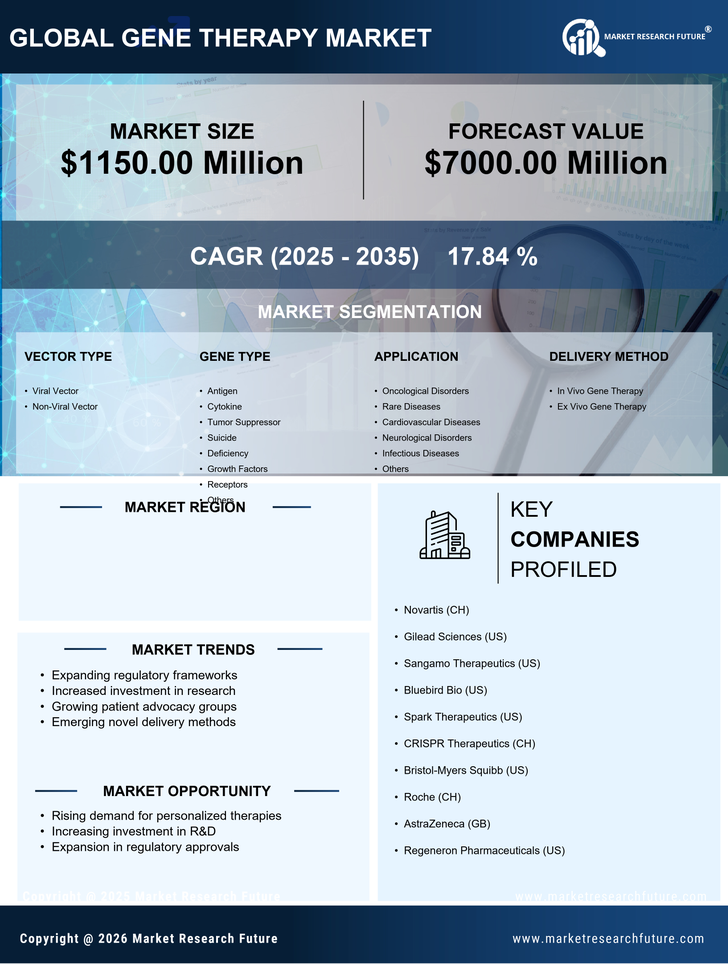
US Chemicals and Materials Market Segmentation
Chemicals and Materials By Vector Type (USD Million, 2025-2035)
- Viral Vector
- Non-Viral Vector
Chemicals and Materials By Gene Type (USD Million, 2025-2035)
- Antigen
- Cytokine
- Tumor Suppressor
- Suicide
- Deficiency
- Growth Factors
- Receptors
- Others
Chemicals and Materials By Application (USD Million, 2025-2035)
- Oncological Disorders
- Rare Diseases
- Cardiovascular Diseases
- Neurological Disorders
- Infectious Diseases
- Others
Chemicals and Materials By Delivery Method (USD Million, 2025-2035)
- In Vivo Gene Therapy
- Ex Vivo Gene Therapy