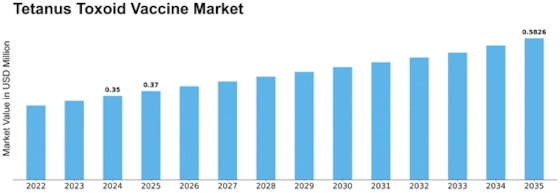
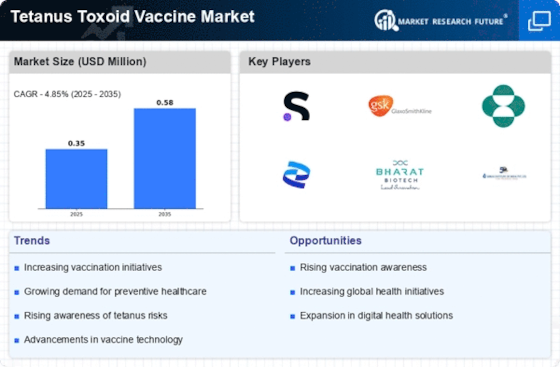
Tetanus Toxoid Vaccine Size
Tetanus Toxoid Vaccine Market Growth Projections and Opportunities
The dynamics and growth trajectory of the Tetanus Toxoid Vaccine market are shaped by a number of factors. The increasing global awareness and focus on immunization programs are among the most significant. Governments and healthcare organizations worldwide are actively promoting vaccination campaigns to prevent tetanus, creating a significant demand for tetanus toxoid vaccines. Furthermore, increased population growth rates in developing countries coupled with an expansion in healthcare infrastructure contributes towards expanding market size.
Another pivotal factor is tetanus cases prevalence as well as effective prevention measures needed. Poorly accessed health facilities maintain Tetanus as a public health burden especially in these areas. High tetanus cases from such locales means that there is need for more vaccines on this disease prompting producers to concentrate mostly on their production and flow into the markets to meet these demands.
Besides, vaccine technology advances play a big role in shaping the Tetanus Toxoid Vaccine market. More efficient and inexpensive vaccines could emerge because of ongoing research and development efforts. Other manufacturing techniques have also been introduced such as use of new adjuvants used during vaccine production process which increases overall efficacy and safety profiles leading to changes market trends for tetanus toxoid vaccines.
Moreover, accessibility and affordability issues affect the Tetanus Toxoid Vaccine market as well. Market expansion is driven by making Vaccines available to a wider range of people including those living in third world nations. These include GAVI, the Vaccine Alliance, and government pharma collaborations aimed at providing cheap vaccines leading to an increase in supply hence increasing their market size.
In addition, regulatory frameworks and approvals significantly impact the Tetanus Toxoid Vaccine market too; stringent health authorities’ regulations; thus affecting manufacturers’ practices together with entry into markets’. This ensures that only quality products are available while building confidence amongst patients or doctors about them.
Tetanus toxoid vaccine’s competitive landscape alongside consolidation also serves as one of the determinants regarding its market dynamics. Presence of key players, mergers and acquisitions as well as strategic alliances bring about market competition. In most cases, companies engage in research and development process to be ahead in the industry thus remaining innovative leading to growth in the aforementioned markets.
Other external factors that affect the Tetanus Toxoid Vaccine market are epidemics and pandemics. The importance of vaccination during global health crises has brought more interest in vaccine discovery and distribution. For instance, the COVID-19 pandemic highlighted the role of a robust healthcare infrastructure and fast-tracked vaccine development efforts thereby indirectly affecting other vaccines such as tetanus toxoid vaccines within its broader spectrum."



















Leave a Comment