Q2 2024: GSK received FDA approval for Benlysta autoinjector for pediatric systemic lupus erythematosus.
Q2 2024: AstraZeneca advanced subcutaneous alirocumab into late-stage trials for SLE AstraZeneca reported the initiation of late-stage clinical trials for its subcutaneous formulation of anifrolumab, aiming to improve patient convenience and adherence in systemic lupus erythematosus treatment.
Q3 2024: FDA granted orphan drug status to multiple CAR-T therapies for lupus erythematosus The FDA announced orphan drug designation for several CAR-T cell therapy candidates targeting systemic lupus erythematosus, reflecting growing regulatory confidence in gene-based approaches for autoimmune diseases.
Q1 2024: GSK reports double-digit growth for Benlysta following expanded indications and autoinjector launch GSK disclosed in its quarterly earnings that Benlysta sales grew at a double-digit rate, driven by expanded indications and the successful launch of its autoinjector format for SLE patients.
Q2 2024: ImmunoACT secured $30M funding for CAR-T therapy development lupus erythematosus ImmunoACT announced the closing of a $30 million funding round to accelerate development and clinical trials of its CAR-T cell therapy targeting systemic lupus erythematosus.
Q2 2025: FDA approves new subcutaneous biologic for systemic lupus erythematosus The FDA granted approval for a new subcutaneous biologic therapy for systemic lupus erythematosus, marking a significant advancement in patient-managed treatment options.
Q1 2025: Biogen partnered with Lupus Research Alliance to develop next-generation SLE therapies. Biogen revealed a strategic partnership with the Lupus Research Alliance to co-develop novel therapies for systemic lupus erythematosus, focusing on biologics and gene-based treatments.
Q3 2024: Roche opens new manufacturing facility for biologics targeting autoimmune diseases including SLE Roche inaugurated a state-of-the-art manufacturing facility dedicated to producing biologic drugs for autoimmune diseases, with a focus on systemic lupus erythematosus therapies.
Q2 2024: Eli Lilly files IND for novel oral therapy for systemic lupus erythematosus Eli Lilly submitted an Investigational New Drug (IND) application to the FDA for a new oral therapy targeting systemic lupus erythematosus, aiming to expand its autoimmune disease portfolio.
Q1 2025: Novartis acquires autoimmune biotech startup focused on SLE gene therapies Novartis acquired a biotech startup focused on SLE gene therapies. Novartis completed the acquisition of a biotech startup specializing in gene therapies for systemic lupus erythematosus, strengthening its pipeline in autoimmune disease treatments.
Q2 2025: FDA updates guidelines to prioritize steroid minimization in SLE treatment The FDA released updated treatment guidelines for systemic lupus erythematosus, emphasizing the minimization of steroid use and encouraging adoption of biologic therapies.
Q3 2024: GSK expands Benlysta manufacturing capacity to meet rising global demand GSK announced the expansion of its Benlysta manufacturing facilities to address increasing global demand for the biologic, following recent regulatory approvals and indication expansions.
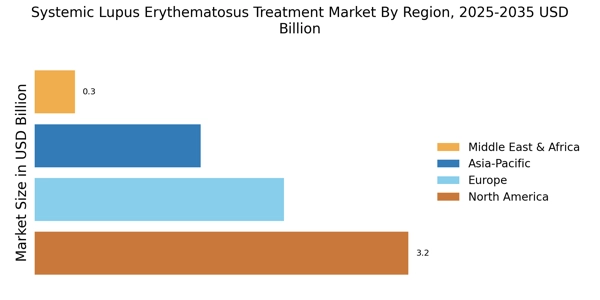
These developments are expected to positively influence the systemic lupus erythematosus treatment market share over the coming years.