ID: MRFR/MED/50950-HCR
200 Pages
Vikita Thakur
Last Updated: February 06, 2026
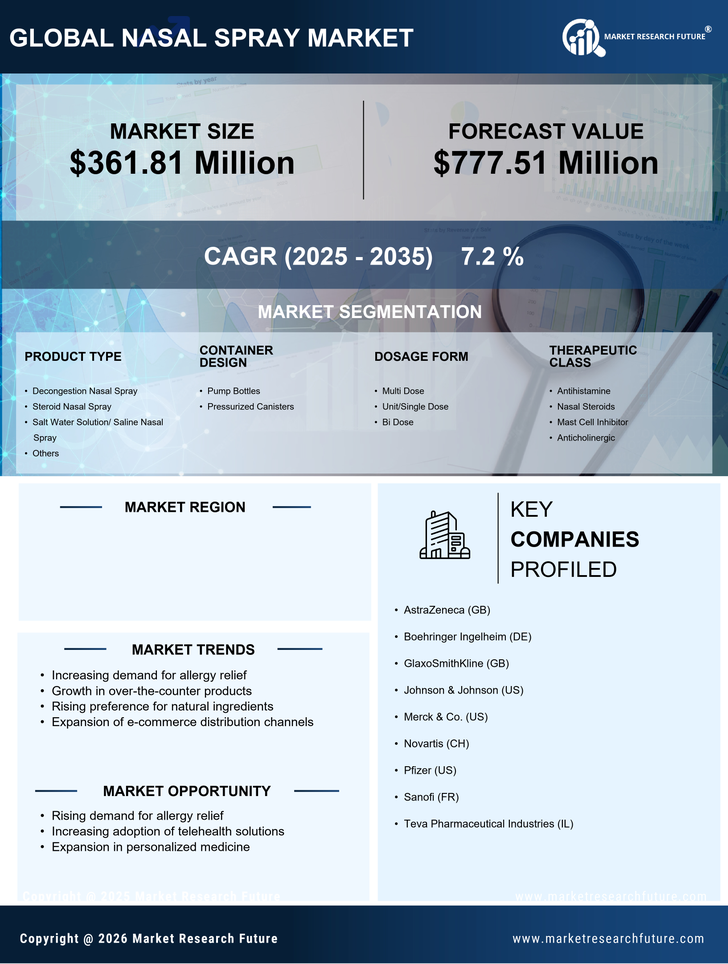
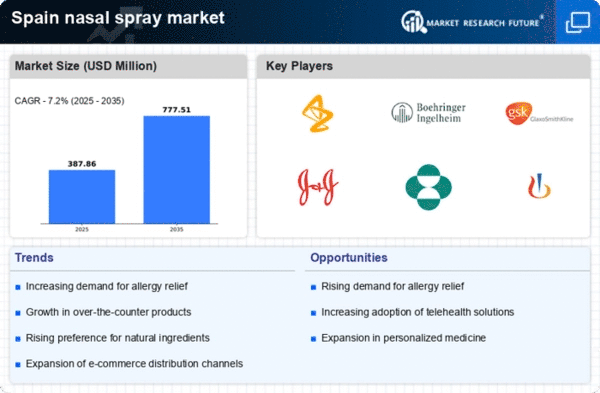
Spain Nasal Spray Market Research Report By Product Type (Decongestion Nasal Spray, Steroid Nasal Spray, Salt Water Solution/ Saline Nasal Spray, Others), By Container Design (Pump Bottles, Pressurized Canisters), By Dosage Form (Multi Dose, Unit/Single Dose, Bi Dose), By Therapeutic Class (Antihistamine, Nasal Steroids, Mast Cell Inhibitor, Anticholinergic), By Application (Nasal Congestion, Allergic And Non-Allergic Rhinitis, Central Nervous System Disorders, Vaccination, Others), By Prescription/Availability (Over The Counter, Prescribed) and By End User (Home Care Settings, Hospitals, Clinics, Community Health Care) - Growth & Industry Forecast 2025 To 2035