Sepsis Diagnostics Market Therapeutics Summary
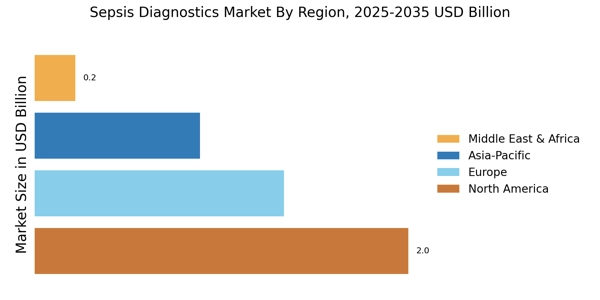
According to Market Research Future analysis, the Sepsis Diagnostics Market Size was estimated at 1.2 USD Billion in 2025. The Sepsis Diagnostics & Therapeutics industry is projected to grow from 1.3 USD Billion in 2026 to 2.7 USD Billion by 2035, exhibiting a compound annual growth rate (CAGR) of 8.3% during the forecast period 2026 - 2035. North America led the market with over 45% share, generating around USD 2.0 billion in revenue.
Rising global sepsis incidence and demand for rapid, accurate diagnosis are key growth drivers in the sepsis diagnostics market. Increasing ICU admissions, early detection needs, and adoption of molecular testing and biomarker-based tools are further accelerating market expansion worldwide.
- According to the World Health Organization (WHO), sepsis affects approximately 49 million people annually and causes around 11 million deaths worldwide, accounting for nearly 20% of global deaths. The Centers for Disease Control and Prevention (CDC) also reports that at least 1.7 million adults in the U.S. develop sepsis each year, highlighting the urgent need for advanced diagnostic solutions to improve survival rates and early intervention outcomes.
Key Market Trends & Highlights
The Sepsis Diagnostics Market is poised for substantial growth driven by technological advancements and increasing awareness.
- Rising sepsis incidence and rapid diagnostic adoption are driving global market expansion across hospitals and laboratories significantly.
- WHO reports approximately 49 million sepsis cases and 11 million deaths annually, highlighting urgent diagnostic demand worldwide.
- Molecular diagnostics dominate with 62% share in 2024 due to superior accuracy and fast pathogen identification capabilities.
- North America leads market with over 45% global revenue share driven by advanced healthcare infrastructure.
- Europe holds 30% share valued at USD 0.36 Billion in 2024, supported by strong regulatory and innovation frameworks.
Market Size & Forecast
| 2025 Market Size | 1.2 (USD Billion) |
| 2035 Market Size | 2.7 (USD Billion) |
| CAGR (2025 - 2035) | 8.3% |
Major Players
Companies such as Thermo Fisher Scientific (US), Roche Diagnostics (CH), Abbott Laboratories (US), Siemens Healthineers (DE), bioMerieux (FR), Becton Dickinson and Company (US), Cepheid (US), Hologic (US), GenMark Diagnostics (US) are some of the major participants in the global market.