North America : Market Leader in Innovation
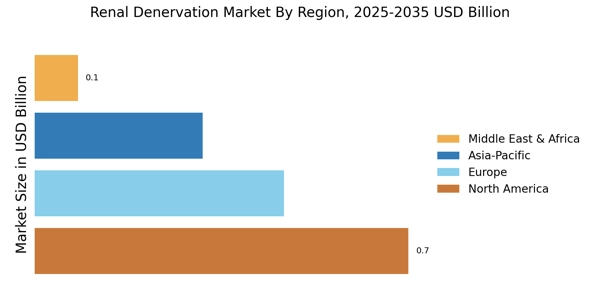
North America leads in the Osteoarthritis Market Size, accounting for over 50.78% of the global revenue in 2024. The region's growth is driven by increasing hypertension prevalence, technological advancements, and supportive regulatory frameworks. The FDA's approval of innovative devices has catalyzed market expansion, with a growing emphasis on minimally invasive procedures. The U.S. is the primary contributor, followed by Canada, which is witnessing rising demand for renal denervation therapies.
The competitive landscape in North America is robust, featuring key players such as Medtronic, Abbott, and Boston Scientific. These companies are investing heavily in R&D to enhance device efficacy and patient outcomes. The presence of advanced healthcare infrastructure and a high rate of adoption of new technologies further bolster market growth. Additionally, partnerships and collaborations among leading firms are expected to drive innovation and expand market reach.
Europe : Emerging Regulatory Frameworks
Europe Osteoarthritis Market was valued at USD 0.46 billion in 2024, making it the second-largest regional market with a 30% share. The region's growth is fueled by increasing awareness of hypertension management and favorable reimbursement policies. Regulatory bodies are actively promoting innovative treatment options, which is expected to enhance market penetration. Countries like Germany and France are leading in adoption rates, supported by strong healthcare systems and increasing patient populations.
The competitive landscape in Europe features significant players such as Terumo Corporation and Astellas Pharma. The market is characterized by a mix of established companies and emerging startups focusing on novel technologies. Collaborative efforts between healthcare providers and manufacturers are fostering innovation, while clinical trials are paving the way for new product approvals. This dynamic environment is expected to sustain growth in the renal denervation market across Europe.
Asia-Pacific : Rapidly Growing Market Potential
Asia-Pacific is an emerging powerhouse in the renal denervation market, holding about 20% of the global share. The region's growth is driven by rising healthcare expenditure, increasing prevalence of hypertension, and a growing aging population. Countries like Japan and Australia are at the forefront, with supportive government initiatives aimed at improving healthcare access and technology adoption. The demand for effective hypertension treatments is expected to surge, further propelling market growth.
The competitive landscape in Asia-Pacific is evolving, with key players like ReCor Medical and Terumo Corporation making significant inroads. The presence of a large patient base and increasing awareness of renal denervation therapies are driving market dynamics. Additionally, collaborations between local firms and international companies are enhancing product offerings and expanding market reach, positioning the region for substantial growth in the coming years.
Middle East and Africa : Untapped Market Opportunities
The Middle East and Africa represent an untapped market for renal denervation, accounting for approximately 5% of the global share. The region's growth is hindered by limited healthcare infrastructure and awareness, but increasing investments in healthcare are paving the way for future expansion. Countries like South Africa and the UAE are beginning to adopt advanced medical technologies, driven by rising healthcare demands and government initiatives to improve health outcomes.
The competitive landscape in this region is still developing, with few key players currently active. However, the presence of multinational companies is expected to increase as the market matures. Local partnerships and collaborations are crucial for enhancing access to renal denervation therapies. As healthcare systems evolve, the potential for growth in the renal denervation market in the Middle East and Africa is significant, with opportunities for innovation and investment.