Market Analysis
In-depth Analysis of Q Fever Market Industry Landscape
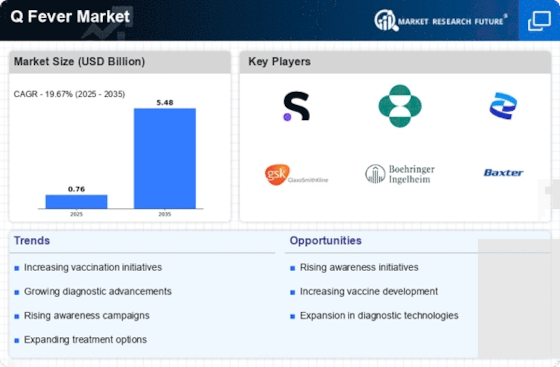
Coxiella burnetii is the bacterium that causes Q fever, a zoonotic infection mainly transmitted from animals to humans. As we delve into the market dynamics of Q Fever, it's crucial to understand the various factors shaping its landscape. The market for Q fever is influenced by both epidemiological and economic considerations. Q Fever incidence varies worldwide with high livestock farming regions having more cases reported. Notably, Australia has been an important epicenter of Q Fever owing to its large scale sheep and cattle husbandry practices.
One of the primary market drivers is increasing awareness and diagnosis of Q Fever. Increasing healthcare awareness leads to more cases being captured and documented. This has boosted demand for diagnostic tools and testing kits leading to increased growth of the Q fever diagnostics market. Furthermore, governments in partnership with health organizations are investing heavily on public health campaigns which educate communities on preventive measures thus expanding the market even further.
Healthcare infrastructure together with regulatory environment play a pivotal role in shaping the dynamics within the Q Fever market. Regions having strong healthcare systems tend to diagnose and manage Q Fever better than most others might be able to do so effectively as well .This directly affects the sector through influencing adoption of sophisticated diagnostic technologies alongside treatment options available .Regulatory frameworks also influence the development as well as commercialization process associated with products related to Q Fevers thereby informing market trends.
Involvement by pharmaceutical industry represents one other significant aspect of these dynamics in this particular trade. Research and development efforts concerning antimicrobial drugs are ongoing despite absence of any specific therapy for q fever at present.The pharmaceutical industry’s interest in addressing unmet medical needs associated with q fever may have substantial effect on market dynamics.Collaborations between pharmaceutical companies, research institutions, and government bodies can accelerate the development of novel therapeutics for Q Fever.
Economic burden of q fever also shapes Market Dynamics since there involves direct and indirect costs such as on healthcare services, productivity losses on one hand and veterinary interventions on the other hand. As a result, there is a growing emphasis on cost-effective preventive measures, such as vaccination programs for at-risk populations and livestock.
Globalization and international trade play a role in the spread of Q Fever and influence market dynamics. The movement of animals and their products can lead to transmission of the bacterium hence affecting how widely q fever occurs in different areas. This interconnectedness underscores the need for collaborative efforts on a global scale to address the challenges posed by Q Fever.
Emerging technologies and innovations also contribute to the market dynamics of Q Fever. Advances in diagnostic tools such as molecular techniques along with serological assays have increased accuracy and efficiency in diagnosing Q fever. Additionally, ongoing research into vaccine development as well as therapeutic modalities can reshape the market landscape providing new avenues for intervention or control in this direction.



















Leave a Comment