Pharmaceutical Suspension Market
ID: MRFR/Pharma/28461-HCR
128 Pages
Rahul Gotadki
Last Updated: April 06, 2026
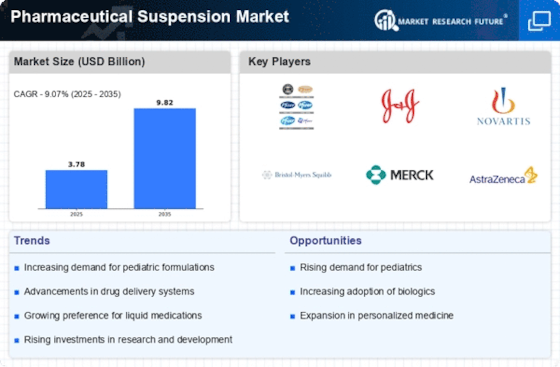
Pharmaceutical Suspension Market Research Report By Dosage Form (Oral Suspension, Injectables, Topical Suspensions, Ophthalmic Suspensions, Others), By Active Ingredient (Antibiotics, Antivirals, Antifungals, Anti-Inflammatories, Analgesics, Antiemetics, Others), By Application (Infectious Diseases, Respiratory Diseases, Pain Management, Gastrointestinal Disorders, Skin Disorders, Ophthalmic Disorders, Others) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035.