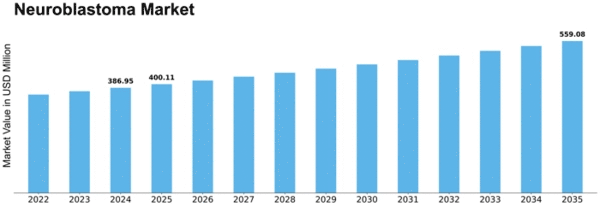
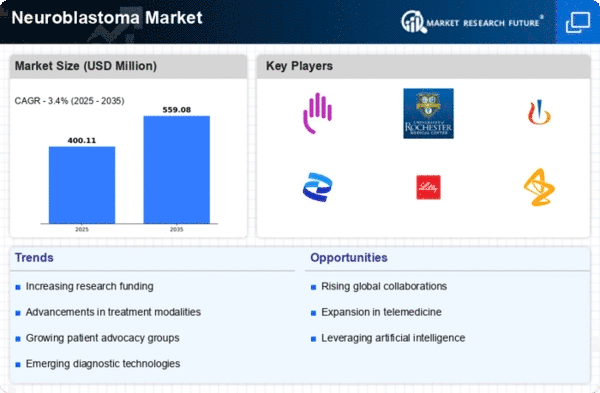
Neuroblastoma Size
Neuroblastoma Market Growth Projections and Opportunities
The Neuroblastoma market is influenced by a range of factors that collectively shape its dynamics and growth trajectory. One of the primary drivers behind the expansion of this market is the prevalence of neuroblastoma, a childhood cancer arising from immature nerve cells. The increasing incidence of neuroblastoma globally has spurred research and development efforts to identify effective treatments, driving the growth of the neuroblastoma market. As awareness about the disease grows and diagnostic capabilities improve, there is an escalating demand for targeted therapies and innovative treatment options.
Technological advancements in medical research and diagnostics significantly impact the neuroblastoma market. Advances in genetic testing, imaging technologies, and molecular profiling have enhanced the understanding of neuroblastoma at a molecular level. This knowledge facilitates the development of precision medicine approaches, leading to more personalized and effective treatment strategies. The integration of cutting-edge technologies into diagnostic and therapeutic processes contributes to the overall progress of the neuroblastoma market.
Regulatory considerations are pivotal in shaping the neuroblastoma market. Stringent regulations govern the approval and commercialization of new drugs and treatments, ensuring their safety and efficacy. The regulatory landscape influences the development timelines of new therapies and shapes the competitive landscape. Changes in regulatory requirements, such as expedited pathways for orphan drug designation, can impact market dynamics by expediting the availability of novel treatments for neuroblastoma.
Economic factors play a crucial role in the neuroblastoma market dynamics. Affordability and accessibility of treatments are influenced by economic conditions, healthcare infrastructure, and insurance coverage. Economic downturns may affect healthcare budgets and patient access to expensive treatments, while economic growth and increased investments in healthcare can positively impact the availability and adoption of advanced neuroblastoma therapies.
Furthermore, the market is influenced by the evolving landscape of oncology research and treatment. Collaborative efforts between academic institutions, pharmaceutical companies, and healthcare organizations contribute to the development of novel therapies for neuroblastoma. The shift towards immunotherapy, targeted therapies, and combination approaches represents a paradigm shift in neuroblastoma treatment strategies, shaping the market's future trajectory.
Competition within the market is another critical factor. The presence of multiple pharmaceutical companies and research institutions fosters innovation and encourages the development of new treatment modalities. Competitive forces drive companies to invest in research, clinical trials, and the development of more effective and targeted neuroblastoma therapies. This competition benefits patients by expanding treatment options and improving overall outcomes.
Market factors are also influenced by awareness and advocacy efforts. As awareness about neuroblastoma increases among healthcare professionals, caregivers, and the general public, there is a growing push for research funding and the development of new therapies. Advocacy organizations play a crucial role in mobilizing support, influencing policy decisions, and fostering collaborations to accelerate progress in the neuroblastoma market.



















Leave a Comment