Market Trends
Key Emerging Trends in the Liver Cirrhosis Treatment Market
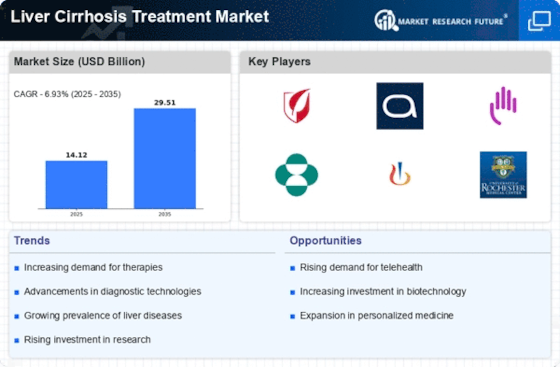
The global liver cirrhosis treatment market is undergoing significant transformations owing to the increased prevalence of liver diseases today. As a result of liver cirrhosis continuing to be a major disease burden, several distinct trends have appeared in its treatment landscape. One notable trend is an increase in early diagnosis and intervention programs worldwide. Additionally, alongside early intervention, there is the emergence of diverse novel therapeutic interventions that look at hepatology issues from another perspective altogether. This has led to pharmaceutical companies putting more effort into discovering new drugs or modes of treatment that can address complex mechanisms underlying the human population's condition towards this deficiency syndrome. The idea behind these discoveries is to improve targeted therapy for patients while avoiding side effects likely to be incurred in most cases. Newer drugs have, therefore, broadened the therapeutic options available, making it possible for doctors to treat patients suffering from hepatocellular carcinoma due to alcohol-related disorders such as hepatitis C virus infection without necessarily resorting to transplantation alone. Also, the current market experiences a higher emphasis on lifestyle changes and holistic approaches toward patient care. This realization has led to the incorporation of comprehensive management strategies that include dietary modifications, exercise routines, and alcohol cessation programs. Integrated care models are gaining traction in the liver cirrhosis treatment market. These models involve collaboration among various healthcare disciplines, including hepatology, gastroenterology, nutrition, and mental health. A multi-disciplinary approach allows for a more comprehensive assessment of patient's needs and facilitates coordinated care, leading to improved patient outcomes and quality of life. Liver disease is a complex condition that requires a multi-disciplinary team approach involving medical professionals from diverse fields who collaborate on behalf of their respective patients. Moreover, there is also an increasing trend in the market's use of non-invasive diagnostic technologies. Consequently, as patients continue to seek alternative methods other than invasive procedures, there are several new non-invasive tools, including elastography and imaging techniques, that have found wide application. Furthermore, efforts to enhance adherence to prescribed therapies by empowering patients with information regarding the disorders' nature as well as available treatment options have led to greater self-management dynamics. Moreover, current market trends show an increased desire for cost-effective treatments. Therefore, with respect to the affordability of effective treatment options, which is a major concern for chronic ailments such as liver cirrhosis, stakeholders are considering ways through which they can make them more accessible. Therefore, this paper shall discuss key factors influencing the development of liver disease treatments in the United States of America today.



















Leave a Comment