Market Analysis
In-depth Analysis of IV Fluid Monitoring Devices Market Industry Landscape
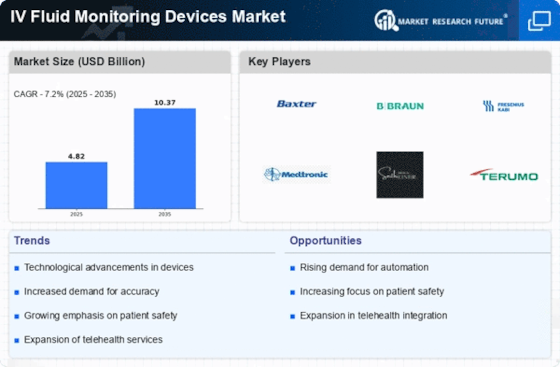
With the increasing prevalence of chronic diseases, surgical procedures, and the aging population, there is a growing demand for IV fluid monitoring devices. These devices play a crucial role in accurately monitoring fluid intake, ensuring patient safety, and preventing complications such as fluid overload or dehydration. Due to the fast-paced developments in the field of IV fluid monitoring apparatus, there have been created more advanced, accurate and sturdy devices which are also more user friendly. Examples like real-time surveillance, wireless connectivity, and network integration with electronic health records further enhance the fluid management efficacy in health-care environments. Examples are the FDA in the USA and the EMA in Europe, who regulate the placement of these devices on the marketplace. Compliance with adherence to the regulative requirements assuming a big role in the opportunity to enter the market and influencing the market dynamics in general. Stringent regulatory requirements that guarantee the safety and performance of these gadgets instead promote their penetration and competitive markets. The IV fluid monitoring devices market has rapidly become the product of competition between the producers of healthcare technology and medical device companies. The market dynamics trend in the direction that is determined by issues such as product invention, pricing tactics, the distribution channel, and line-up of strategic collaborations. Firms try to make their products distinctive trough technical superiority and easier usability and to be able to compete against the main rivals in the market. Inventories of IV fluid monitoring devices are shaped by existing clinical guidelines and standards set by professional medical associations and regulatory bodies as they give directions on when and how these devices should be used in a clinical practice. Compliance to guideline recommendations by evidence-based processes will lead in market dynamics by creating demand for devices that go in line with the clinical guidelines and standards. The economy aspects which incorporate the healthcare spending, reimbursement policy and financial constraints become the chief uncertainty factors for the adoption and utilization of the IV fluid monitoring devices in the healthcare facilities. Efficiency considerations related effort expended to investments and return of investments provide guidance in purchasing of equipment and services by healthcare providers, thus fashioning market environment and market penetration.
The IV fluid monitoring devices market offers opportunities for expansion and growth through the introduction of innovative products, strategic partnerships, and market expansion into emerging regions. Market dynamics are influenced by factors such as market entry barriers, regulatory requirements, and competitive landscape in target markets. The IV fluid monitoring devices market is expected to witness continued growth and innovation driven by advancements in technology, increasing demand for patient safety and quality of care, and the growing adoption of digital health solutions in healthcare settings. Collaboration between stakeholders, investment in research and development efforts, and regulatory support will play a crucial role in shaping the future dynamics of the IV fluid monitoring devices market.



















Leave a Comment