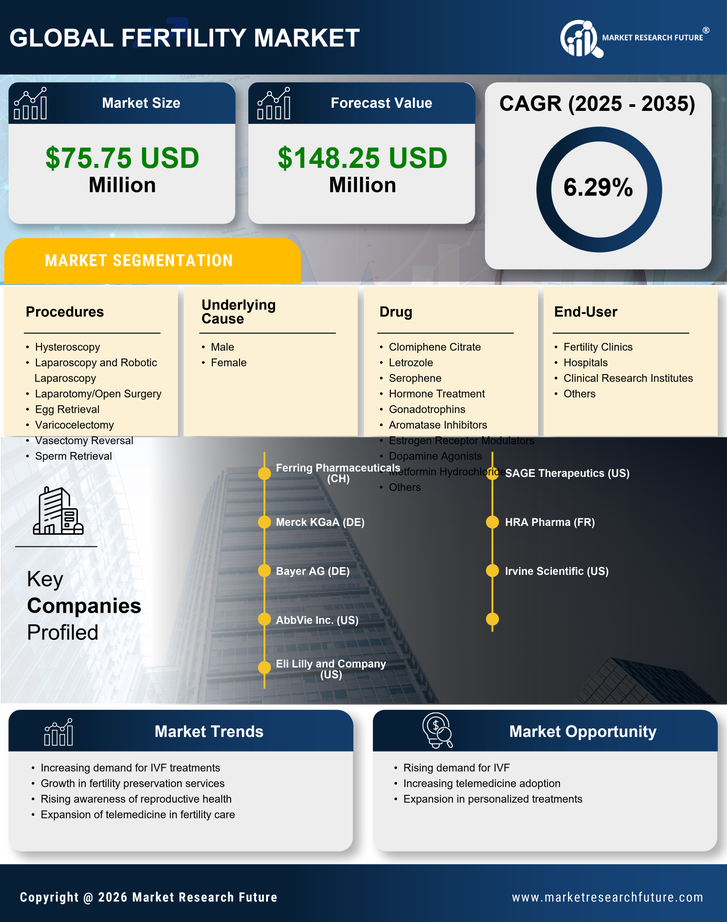
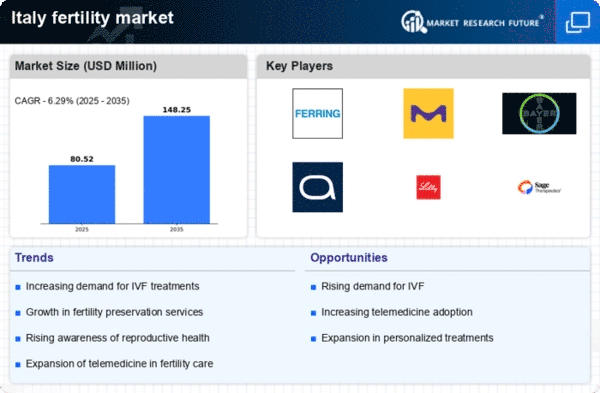
Italy Life Sciences Market Segmentation
Life Sciences By Underlying Cause (USD Million, 2025-2035)
- Male
- Female
Life Sciences By Drug (USD Million, 2025-2035)
- Clomiphene Citrate
- Letrozole
- Serophene
- Hormone Treatment
- Gonadotrophins
- Aromatase Inhibitors
- Estrogen Receptor Modulators
- Dopamine Agonists
- Metformin Hydrochloride
- Others
Life Sciences By Procedures (USD Million, 2025-2035)
- Hysteroscopy
- Laparoscopy and Robotic Laparoscopy
- Laparotomy/Open Surgery
- Egg Retrieval
- Varicocelectomy
- Vasectomy Reversal
- Sperm Retrieval
Life Sciences By End-User (USD Million, 2025-2035)
- Fertility Clinics
- Hospitals
- Clinical Research Institutes
- Others