

- Global Market Outlook
- In-depth analysis of global and regional trends
- Analyze and identify the major players in the market, their market share, key developments, etc.
- To understand the capability of the major players based on products offered, financials, and strategies.
- Identify disrupting products, companies, and trends.
- To identify opportunities in the market.
- Analyze the key challenges in the market.
- Analyze the regional penetration of players, products, and services in the market.
- Comparison of major players’ financial performance.
- Evaluate strategies adopted by major players.
- Recommendations
- Vigorous research methodologies for specific market.
- Knowledge partners across the globe
- Large network of partner consultants.
- Ever-increasing/ Escalating data base with quarterly monitoring of various markets
- Trusted by fortune 500 companies/startups/ universities/organizations
- Large database of 5000+ markets reports.
- Effective and prompt pre- and post-sales support.
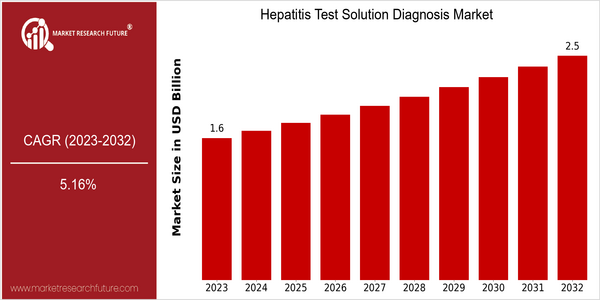
Market Size Snapshot
| Year | Value |
|---|---|
| 2023 | USD 1.59 Billion |
| 2032 | USD 2.5 Billion |
| CAGR (2024-2032) | 5.16 % |
Note – Market size depicts the revenue generated over the financial year
The Hepatitis Test Solution Diagnosis Market is currently valued at approximately USD 1.59 billion in 2023 and is projected to reach USD 2.5 billion by 2032, reflecting a compound annual growth rate (CAGR) of 5.16% from 2024 to 2032. This growth trajectory indicates a steady increase in demand for hepatitis testing solutions, driven by rising awareness of hepatitis infections, advancements in diagnostic technologies, and an increasing focus on preventive healthcare measures globally. As healthcare systems evolve, the integration of innovative testing methods, such as point-of-care testing and molecular diagnostics, is enhancing the accuracy and speed of hepatitis diagnosis, further propelling market expansion. Key players in the hepatitis testing market, including Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers, are actively investing in research and development to introduce more efficient and user-friendly testing solutions. Strategic initiatives such as partnerships with healthcare providers and investments in digital health technologies are also contributing to market growth. For instance, collaborations aimed at improving access to hepatitis testing in underserved regions are expected to play a crucial role in expanding the market footprint. Overall, the combination of technological advancements and strategic initiatives by leading companies is set to drive the Hepatitis Test Solution Diagnosis Market towards significant growth in the coming years.
Regional Market Size
Regional Deep Dive
The Hepatitis Test Solution Diagnosis Market is experiencing significant growth across various regions, driven by increasing awareness of hepatitis infections, advancements in diagnostic technologies, and supportive government initiatives. In North America, the market is characterized by high healthcare expenditure and a robust regulatory framework, while Europe benefits from stringent health policies and a strong emphasis on preventive healthcare. The Asia-Pacific region is witnessing rapid growth due to rising infection rates and improving healthcare infrastructure, whereas the Middle East and Africa face challenges related to access and awareness. Latin America is gradually enhancing its diagnostic capabilities, influenced by both public health initiatives and private sector investments.
Europe
- The European Union has implemented new regulations aimed at standardizing hepatitis testing across member states, which is expected to improve the quality and reliability of diagnostic solutions.
- Innovative partnerships between public health organizations and private companies, such as the collaboration between the World Health Organization (WHO) and various biotech firms, are driving the development of more effective and affordable testing solutions.
Asia Pacific
- Countries like India and China are investing heavily in healthcare infrastructure, leading to increased availability of hepatitis testing solutions, which is crucial for managing the high prevalence of hepatitis infections in these regions.
- Recent initiatives by the World Health Organization to eliminate viral hepatitis as a public health threat by 2030 are prompting governments to enhance their testing capabilities and public awareness campaigns.
Latin America
- Brazil has initiated a national program to increase hepatitis testing and treatment, which is expected to enhance the overall diagnosis rates and reduce the burden of the disease.
- Collaborations between local governments and international health organizations are focusing on improving laboratory capacities and training healthcare professionals in hepatitis diagnostics.
North America
- The U.S. Food and Drug Administration (FDA) has recently approved several rapid hepatitis test kits, enhancing accessibility and speed of diagnosis, which is expected to significantly improve early detection rates.
- Organizations like the Centers for Disease Control and Prevention (CDC) are actively promoting hepatitis screening programs, particularly among high-risk populations, which is likely to increase testing rates and awareness.
Middle East And Africa
- The African Union has launched a campaign to increase hepatitis awareness and testing, which is expected to significantly impact the diagnosis rates in the region, particularly in sub-Saharan Africa.
- Local companies are beginning to develop low-cost hepatitis test kits tailored to the needs of the region, which could improve access to testing in underserved areas.
Did You Know?
“Approximately 290 million people worldwide are unaware they are infected with hepatitis B, highlighting the critical need for improved testing and awareness initiatives.” — World Health Organization (WHO)
Segmental Market Size
The Hepatitis Test Solution Diagnosis Market is currently experiencing stable growth, driven by increasing awareness of hepatitis infections and the need for early diagnosis. Key factors propelling demand include rising global prevalence rates of hepatitis B and C, alongside stringent regulatory policies aimed at improving screening and treatment protocols. Technological advancements in diagnostic methods, such as rapid testing and point-of-care solutions, further enhance accessibility and efficiency in testing. Currently, the market is in a mature adoption stage, with notable players like Abbott Laboratories and Roche leading the way in innovative testing solutions. Primary applications include routine screening in healthcare settings, blood donation centers, and high-risk population assessments. Trends such as the ongoing emphasis on public health initiatives and government mandates for hepatitis screening are catalyzing growth. Additionally, the integration of digital health technologies, including telemedicine and mobile health applications, is shaping the segment's evolution, making testing more convenient and widespread.
Future Outlook
The Hepatitis Test Solution Diagnosis Market is poised for significant growth from 2023 to 2032, with a projected market value increase from $1.59 billion to $2.5 billion, reflecting a compound annual growth rate (CAGR) of 5.16%. This growth trajectory is driven by an increasing prevalence of hepatitis infections globally, heightened awareness of viral hepatitis, and the implementation of robust screening programs by governments and health organizations. As more individuals gain access to healthcare services, particularly in developing regions, the demand for hepatitis testing solutions is expected to rise, leading to greater market penetration and usage rates. Technological advancements in diagnostic methods, such as the development of rapid testing kits and point-of-care testing solutions, are anticipated to further enhance market growth. These innovations not only improve the accuracy and speed of diagnosis but also facilitate early detection and treatment, which are critical in managing hepatitis infections. Additionally, supportive policies and funding from international health bodies, such as the World Health Organization, aimed at eliminating viral hepatitis as a public health threat by 2030, will likely bolster market expansion. Emerging trends, including the integration of digital health technologies and telemedicine in hepatitis testing, are expected to reshape the landscape, making testing more accessible and efficient for patients worldwide.
Covered Aspects:| Report Attribute/Metric | Details |
|---|---|
| Growth Rate | 3.86% (2022-2030) |
Hepatitis Test Solution Diagnosis Market Highlights:
Leading companies partner with us for data-driven Insights
Kindly complete the form below to receive a free sample of this Report
Tailored for You
- Dedicated Research on any specifics segment or region.
- Focused Research on specific players in the market.
- Custom Report based only on your requirements.
- Flexibility to add or subtract any chapter in the study.
- Historic data from 2014 and forecasts outlook till 2040.
- Flexibility of providing data/insights in formats (PDF, PPT, Excel).
- Provide cross segmentation in applicable scenario/markets.









