Market Trends
Key Emerging Trends in the Hepatitis C Drugs Market
New developments in Direct-Acting Antivirals (DAAs) have a big effect on the market for Hepatitis C drugs. These antiviral drugs work on specific stages of the HCV lifecycle. Compared to traditional interferon-based medicines, they have higher cure rates, faster treatment times, and fewer side effects.
Pan-genotypic treatments for Hepatitis C are getting a lot of attention. The creation of drugs that work against various HCV genotypes makes treatment easier to get, helps doctors make decisions about treatment, and handles the wide range of Hepatitis C strains found around the world.
Treatment access programs are making the market for hepatitis C drugs grow. Pharmaceutical companies, states, and non-profits are working to make it easier for people to get cheap Hepatitis C medicines. This helps more people get treatment, especially in low- and middle-income countries.
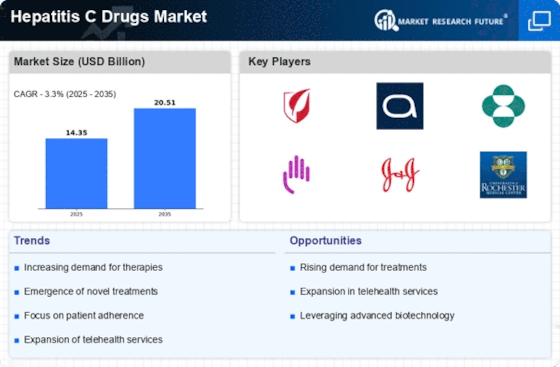
Oral versions and treatment plans that are easier to follow are becoming more popular. Interferon shots are no longer needed with all-oral methods. This makes it easier for patients to stick with their treatment, lowers side effects, and makes Hepatitis C treatment more tolerable overall.
Generic and biosimilar drugs are becoming more and more mixed in the market. Generic forms of DAAs help bring down the price of medicines, which makes treatment for Hepatitis C easier for more people and encourages competition in the market.
There is a clear focus on certain groups, such as people who have more than one illness, like HIV or cirrhosis of the liver. Adapting treatment plans to the specific needs of each patient improves the effectiveness of therapy and solves the unique problems that come up when managing Hepatitis C in different groups.
Hepatitis C removal attempts around the world are changing how markets work. The World Health Organization wants to get rid of Hepatitis C as a public health threat by 2030. This goal drives study, growth, and access programs, which encourages people to work together to fight the disease.
In the market for Hepatitis C drugs, patient education and lobbying efforts are very important. Programs that raise knowledge, lower shame, and give people information help with early identification, starting treatment, and having better results. The main goal of study is to make the next wave of antiviral drugs. Drug resistance and the need for new treatments are two problems that new medicines with different ways of working are trying to solve. These new medicines include host-targeted therapies and new mixtures.
Point-of-care testing is becoming more important for managing Hepatitis C. Rapid testing tools help find HCV cases quickly and accurately, so treatment can begin quickly and effectively, which improves the general quality of care for people with Hepatitis C.
The market is changing because more and more people are using telemedicine and online tracking. Telehealth systems allow for online talks, tracking of drug following, and follow-up care. This is especially helpful for patients in rural or underdeveloped areas.



















Leave a Comment