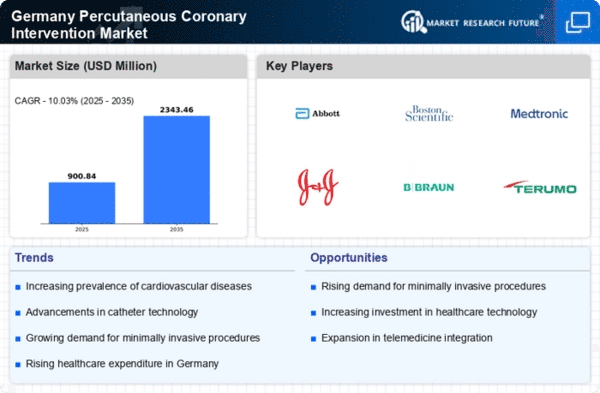
Germany Pharmaceutical Market Segmentation
Pharmaceutical By Product Type (USD Million, 2025-2035)
- Coronary Stents
- Balloon Catheters
- Coronary Guidewires
- Guiding Catheters
- Guiding Sheaths
- Diagnostic Catheters
- Crossing Catheters
- Inflation Device
- Guidance System
- Peripheral Thrombectomy System
- Embolic Protection System
- Accessories
Pharmaceutical By Vascular Access (USD Million, 2025-2035)
- Radial
- Femoral
Pharmaceutical By End User (USD Million, 2025-2035)
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Others