Gene Therapy Clinical Trial Service Market
ID: MRFR/Pharma/27814-HCR
128 Pages
Rahul Gotadki
Last Updated: April 06, 2026
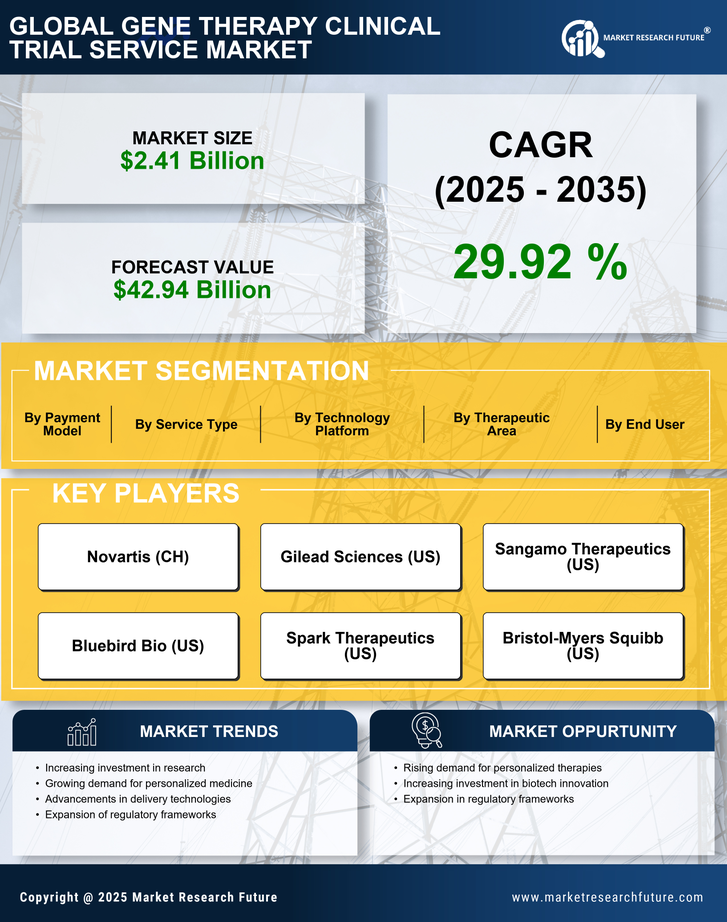
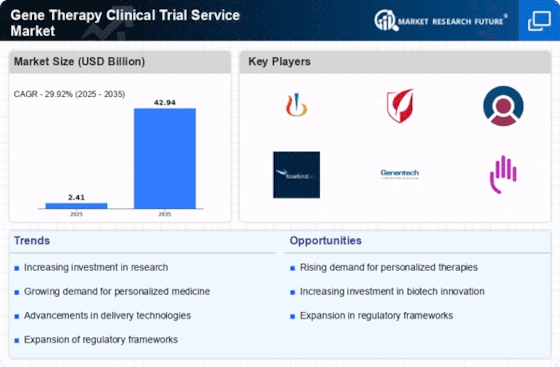
Gene Therapy Clinical Trial Service Market Research Report By Service Type (Preclinical Services, Clinical Trial Management, Data Management and Analysis, Regulatory Affairs and Submissions, Quality Assurance and Control, Post-Marketing Surveillance), By Therapeutic Area (Oncology, Rare Diseases, Cardiovascular Diseases, Neurological Disorders, Infectious Diseases, Metabolic Disorders), By Technology Platform (Lentiviral Vectors, Retroviral Vectors, Adeno-Associated Viral (AAV) Vectors, CRISPR-Cas9, Gene Editing, Oligonucleotide Therapies), By Payment Model (Fee-for-Service, Value-Based Pricing, Subscription-Based Model, Risk-Sharing Agreements, Milestone Payments), By End User (Pharmaceutical Companies, Biotechnology Companies, Academic Institutions, Government Research Institutes, Contract Research Organizations (CROs)) and By Regional (North America, Europe, South America, Asia Pacific, Middle East and Africa) - Growth & Industry Forecast 2025 To 2035