Market Share
Fungal Endocarditis Market Share Analysis
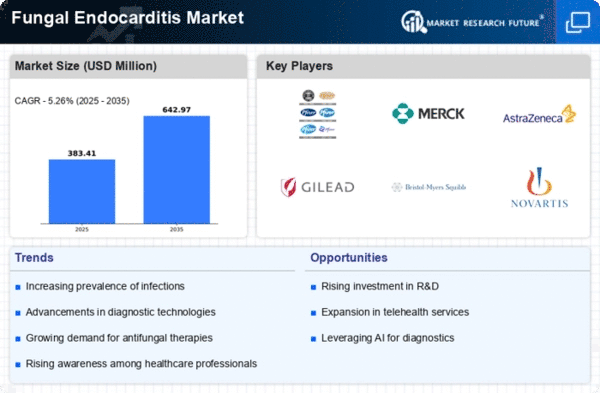
The Fungal Endocarditis Market tends to a difficult ailment described by fungal contaminations influencing the endocardium. Organizations working in this specialty market utilize different techniques to situate themselves really and capture market share in the finding and treatment of fungal endocarditis. A key procedure includes the usage of cutting-edge indicative strategies. Organizations put resources into cutting edge imaging innovations, blood culture techniques, and molecular diagnostics to improve the exactness and speed of fungal endocarditis determination. Remaining at the very front of analytic abilities adds to market authority. Given the intricacy of treating fungal endocarditis, organizations decisively center around the turn of events and advancement of compelling antifungal treatments. This incorporates the exploration and presentation of novel antifungal specialists and treatment conventions. Offering exhaustive and inventive treatment choices improves market appeal and impacts endorsing choices. Dynamic commitment to instructive projects focusing on the two patients and healthcare experts is indispensable. Giving data about fungal endocarditis, its risk elements, side effects, and available medicines upgrades consciousness. Instructing doctors on the subtleties of diagnosing and overseeing fungal endocarditis adds to expanded reception and market share development. A contemporary procedure includes coordinating fungal endocarditis determination and conferences with telemedicine stages. Organizations foster arrangements that permit remote interviews with irresistible infection trained professionals, empowering patients to get to well-qualified feelings paying attention to geographical constraints. This approach lines up with the developing pattern of telehealth and works on tolerant accessibility. Giving expense proficient antifungal treatment arrangements is an essential thought. Organizations upgrade fabricating processes, haggle with providers, and investigate economies of scale to offer serious valuing. This system draws in healthcare suppliers and establishments searching for solid and practical antifungal treatments for fungal endocarditis.



















Leave a Comment