COVID 19 Diagnostics Market Summary
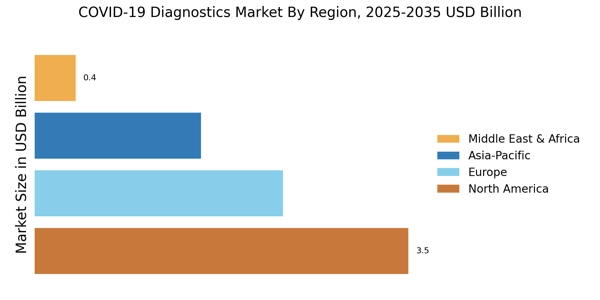
The Global COVID-19 Diagnostics Market size was valued at USD 7.876 Billion in 2024, and the market is projected to grow from USD 8.184 Billion in 2025 to USD 12.01 Billion by 2035, registering a CAGR of 3.91% during the forecast period 2025–2035. North America led the market in 2024 with over 44.44% share, generating around USD 3.5 Billion in revenue.
Rising global demand for rapid and accurate disease detection is driving the COVID-19 diagnostics market. Increased testing initiatives, early diagnosis needs, and strengthened healthcare preparedness are boosting adoption of molecular and antigen-based diagnostic solutions across healthcare systems worldwide.
According to the World Health Organization (WHO) Data Portal, over 13.1 billion COVID-19 vaccine doses were administered globally alongside extensive testing programs, with countries reporting millions of diagnostic tests weekly during peak waves, reinforcing sustained demand for scalable and rapid diagnostic infrastructure and supporting continuous market expansion.
Key Market Trends & Highlights
The COVID-19 Diagnostics Market is experiencing dynamic growth driven by technological advancements and evolving healthcare needs.
- North America leads with over 44.44% share, supported by advanced healthcare systems and high diagnostic testing volumes.
- Europe holds 30% share, valued at USD 2.26 Billion in 2024, making it second-largest regional market.
- PCR Tests dominate with 68% share, driven by high accuracy and widespread clinical adoption across healthcare systems.
- Hospitals account for 55% share, while direct sales lead distribution with 58%, ensuring institutional procurement dominance globally.
Market Size & Forecast
| 2024 Market Size | 7.876 (USD Billion) |
| 2035 Market Size | 12.01 (USD Billion) |
| CAGR (2025 - 2035) | 3.91% |
Major Players
Companies such as Abbott Laboratories (US), Roche Diagnostics (CH), Thermo Fisher Scientific (US), Siemens Healthineers (DE), BD (US), Cepheid (US), Hologic (US), PerkinElmer (US), Bio-Rad Laboratories (US) are some of the major participants in the global market.