Contract Research Organization Market Summary
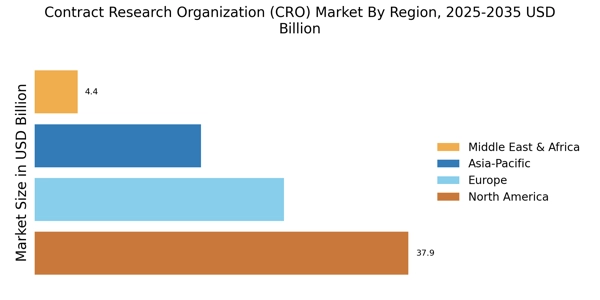
As per Market Research Future Reports analysis, the Contract Research Organization Market size was valued at USD 84.3 Billion in 2024. The market is projected to grow from USD 89.88 Billion in 2025 to USD 170.63 Billion by 2035, exhibiting a CAGR of 6.62% during the forecast period 2025–2035. North America led the market with over 44.96% share, generating around USD 37.9 billion in revenue.
The Contract Research Organization (CRO) market is primarily driven by the rising global burden of chronic diseases and increasing demand for efficient drug development, encouraging pharmaceutical companies to outsource clinical research to specialized organizations for faster, cost-effective, and compliant trial execution.
According to the World Health Organization (WHO), noncommunicable diseases account for 74% of global deaths annually (41 million people), significantly increasing demand for clinical trials and accelerating reliance on CROs to manage expanding research pipelines efficiently.
Key Market Trends & Highlights
The Contract Research Organization Market is experiencing robust growth driven by technological advancements and increased outsourcing.
- North America leads with 44.96% share in 2024, driven by strong clinical research infrastructure.
- Europe accounts for 25% share, valued at USD 21.075 billion in 2024.
- Clinical trials dominate with 69% share, driven by increasing global R&D investments and complex drug development pipelines.
- Pharmaceutical companies hold 65% share, supported by large-scale outsourcing strategies and rising therapeutic innovation globally.
Market Size & Forecast
| 2024 Market Size | 84.3 (USD Billion) |
| 2035 Market Size | 170.63 (USD Billion) |
| CAGR (2025 - 2035) | 6.62% |
Major Players
Companies such as IQVIA (US), Labcorp Drug Development (US), PPD (US), Syneos Health (US), Charles River Laboratories (US), Medpace (US), PRA Health Sciences (US), Wuxi AppTec (CN), Celerion (US) are some of the major participants in the global market.