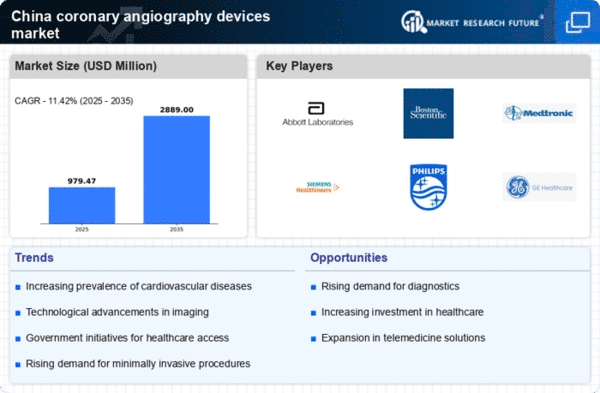
China Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Angiography Catheters
- Contrast Media
- Angiography Systems
- Inflatable Balloons
- Guidewires
Medical Device By Application (USD Million, 2025-2035)
- Coronary Angiography
- Peripheral Angiography
- Neuro Angiography
- Interventional Angiography
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Ambulatory Surgical Centers
- Diagnostic Imaging Centers
- Specialty Clinics
Medical Device By Technology (USD Million, 2025-2035)
- Digital Angiography
- Computed Tomography Angiography
- Magnetic Resonance Angiography