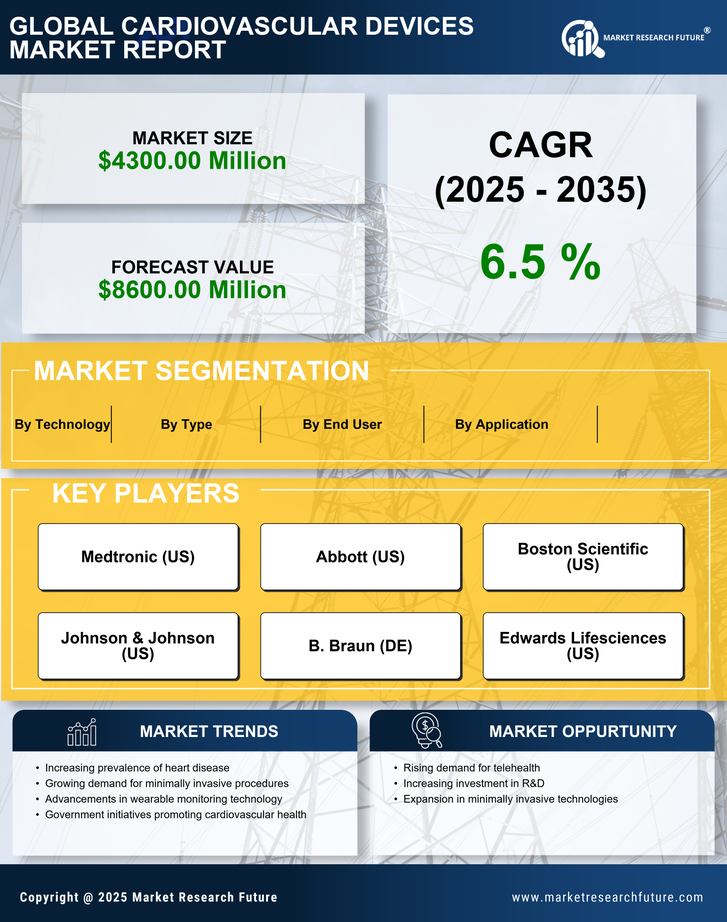
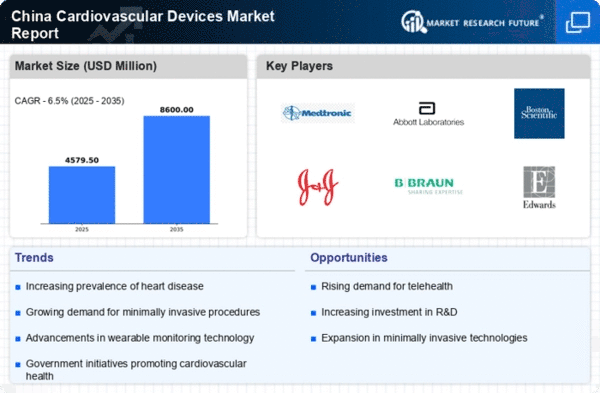
China Medical Device Market Segmentation
Medical Device By Type (USD Million, 2025-2035)
- Diagnostic Devices
- Monitoring Devices
- Therapeutic Devices
- Surgical Devices
Medical Device By Application (USD Million, 2025-2035)
- Coronary Artery Disease
- Heart Failure
- Arrhythmia
- Valvular Heart Disease
Medical Device By End User (USD Million, 2025-2035)
- Hospitals
- Cardiac Centers
- Ambulatory Surgical Centers
- Home Healthcare
Medical Device By Technology (USD Million, 2025-2035)
- Implantable Pacemakers
- Cardiac Stents
- Ablation Devices
- Heart Valve Devices