Cell Line Development Market Summary
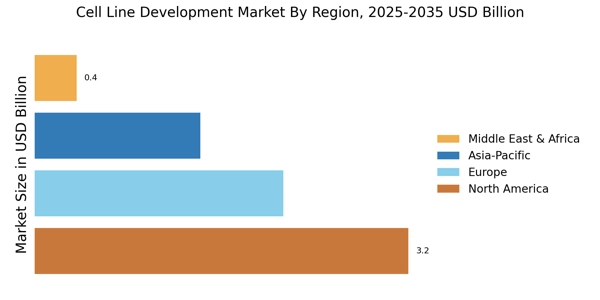
According to Market Research Future analysis, the Cell Line Development Market size was valued at USD 7.11 Billion in 2024. The market is projected to grow from USD 7.972 Billion in 2025 to USD 25.03 Billion by 2035, exhibiting a CAGR of 12.1% during the forecast period 2025-2035. North America led the market with over 45% share, generating around USD 3.2 billion in revenue.
Rising demand for biologics and advanced therapeutic development is driving cell line development adoption, supported by expanding monoclonal antibody production, vaccine innovation, and personalized medicine approaches that require stable, high-yield, and reproducible cell systems for efficient drug discovery and manufacturing.
According to WHO, noncommunicable diseases cause 41 million deaths annually, representing 74% of global mortality, increasing demand for biologics and advanced therapies. Growing therapeutic needs, including vaccines and monoclonal antibodies, directly support scalable cell line development for improved global treatment access and biomanufacturing capacity.
Key Market Trends & Highlights
The Cell Line Development Market is poised for substantial growth driven by technological advancements and increasing demand for biopharmaceuticals.
- North America leads with over 45% share in 2024, supported by strong biopharmaceutical R&D and chronic disease burden.
- Europe holds 30% market share in 2024, valued at USD 2.13 billion, driven by strong regulatory healthcare frameworks.
- Drug discovery segment dominated with 52% share in 2024, fueled by rising global noncommunicable disease burden and innovation.
- Hybridoma cell lines accounted for 61% share in 2024, driven by global monoclonal antibody demand for cancer treatment therapies.
Market Size & Forecast
| 2024 Market Size | 7.11 (USD Billion) |
| 2035 Market Size | 25.03 (USD Billion) |
| CAGR (2025 - 2035) | 12.12% |
Major Players
Companies such as Thermo Fisher Scientific (US), Merck KGaA (DE), Lonza Group (CH), GE Healthcare (US), Sartorius AG (DE), WuXi AppTec (CN), Celerion (US), Charles River Laboratories (US), Sigma-Aldrich (US) are some of the major participants in the global market.