Cancer Immunotherapy Market Summary
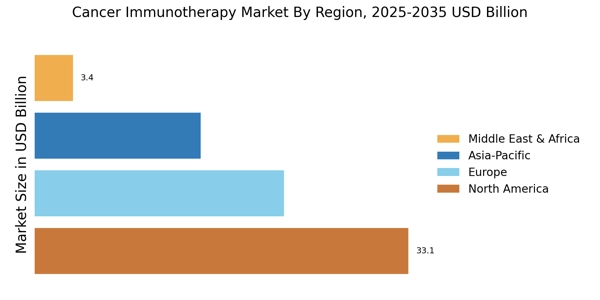
The Global Cancer Immunotherapy Market size was valued at USD 73.7 Billion in 2024, and the market is projected to grow from USD 77.38 Billion in 2025 to USD 125.93 Billion by 2035, registering a CAGR of 4.99% during the forecast period 2025–2035. North America led the market with over 42.78% share, generating around USD 33.1 billion in revenue.
The rising global cancer burden and increasing adoption of personalized therapies are major growth drivers for the Cancer Immunotherapy Market. Expanding investments in immuno-oncology research, combined with advancements in targeted immune therapies, continue accelerating demand for innovative cancer treatment solutions worldwide.
According to the World Health Organization, cancer caused nearly 10 million deaths globally in 2022, making it a leading cause of mortality. Growing incidence rates and increasing focus on precision oncology are significantly strengthening demand for advanced immunotherapy solutions and personalized cancer treatments worldwide.
Key Market Trends & Highlights
The Cancer Immunotherapy Market is experiencing robust growth driven by innovative therapies and increasing demand for personalized treatment options.
- North America led the Cancer Immunotherapy Market in 2024, contributing over 42.78% of total global revenue.
- Europe Cancer Immunotherapy Market reached USD 20.5 billion in 2024, representing approximately 26.49% global share.
- Monoclonal antibodies dominated with 44% share, driven by strong efficacy across multiple oncology indications.
- Intravenous administration accounted for 58% market share due to rapid delivery and widespread clinical use.
Market Size & Forecast
| 2024 Market Size | 73.7 (USD Billion) |
| 2035 Market Size | 125.93 (USD Billion) |
| CAGR (2025 - 2035) | 4.99% |
Major Players
Companies such as Bristol-Myers Squibb (US), Merck & Co. (US), Roche (CH), Novartis (CH), Amgen (US), AstraZeneca (GB), Gilead Sciences (US), Regeneron Pharmaceuticals (US), Eli Lilly and Company (US) are some of the major participants in the market.