Biosensors Market Summary
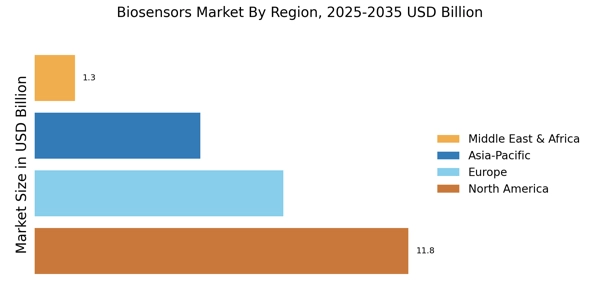
According to MRFR analysis, the Biosensors Market Size was valued at USD 26.22 Billion in 2024. The market is projected to grow from USD 28.25 Billion in 2025 to USD 59.43 Billion by 2035, registering a CAGR of 7.72% during the forecast period (2025–2035). North America led the market with over 40.05% share, generating around USD 10.5 billion in revenue.
The Biosensors Market is growing rapidly due to increasing demand for rapid diagnostics, rising prevalence of chronic diseases, and expanding use of wearable health monitoring devices. Key trends include advancements in nanotechnology-based sensors, integration with digital health platforms, and growing applications in medical diagnostics, environmental monitoring, and food safety.
Key Market Trends & Highlights
The Biosensors Market is experiencing robust growth driven by technological advancements and increasing demand for point-of-care testing.
- North America remains the largest market for biosensors, driven by advanced healthcare infrastructure and innovation.
- The Asia-Pacific region is the fastest-growing market, fueled by rising healthcare investments and a focus on health and wellness.
- The healthcare segment dominates the market, while the food safety segment is emerging rapidly due to heightened safety standards.
- Technological innovations in biosensors and the rising prevalence of chronic diseases are key drivers propelling market expansion.
Market Size & Forecast
| 2024 Market Size | 26.22 (USD Billion) |
| 2035 Market Size | 59.43 (USD Billion) |
| CAGR (2025 - 2035) | 7.72% |
Major Players
Companies such as Abbott Laboratories (US), Roche Diagnostics (CH), Siemens Healthineers (DE), Medtronic (US), Thermo Fisher Scientific (US), Johnson & Johnson (US), Biosense Webster (US), Dexcom (US), Acon Laboratories (US) are some of the major participants in the global market.