Market Analysis
In-depth Analysis of Autoimmune Disease Diagnostics Market Industry Landscape
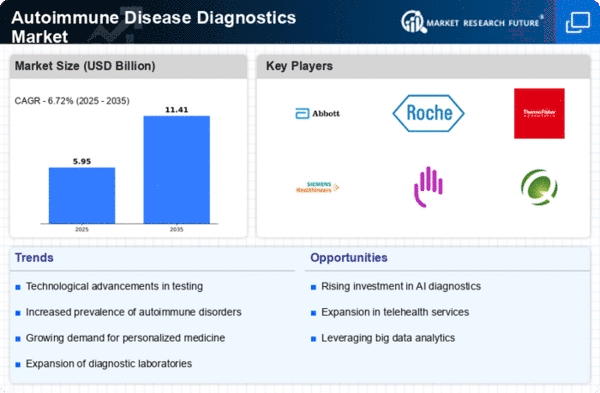
The increasing prevalence of autoimmune diseases worldwide has led to a considerable surge in the market for autoimmune disease diagnostics in recent times. The market has grown significantly in tandem with increased awareness of these illnesses, which has increased demand for precise and reliable indicator devices. Innovative approaches, including as biomarker identification, subatomic diagnostics, and advanced imaging techniques, have improved the accuracy and quickness of autoimmune disease detection, hence attracting a larger patient population. An upsurge in autoimmune disorders, such as multiple sclerosis, lupus, and rheumatoid arthritis, has increased demand for sound analytical frameworks. Natural causes, lifestyle modifications, and genetic predispositions all contribute to the escalating occurrence of these issues, which fuels market interest. A variety of demonstrative testing phases, such as in vitro diagnostics, imaging methods, and serological assays, are used to characterize the market. These phases address different autoimmune conditions, allowing healthcare providers to customize their recommended approaches based on the specific needs of their clients. Spending more on healthcare, particularly in developed areas, has made it possible to access state-of-the-art diagnostics for autoimmune diseases. The availability and affordability of demonstrative tools improve when states and private sectors allocate more funds to the medical services system, spurring market growth. One major motivator has been helping patients become more cognizant of autoimmune illnesses and the importance of early detection. People are searching for appropriate clinical mediation at this time due to expanded education and mindfulness campaigns, which is contributing to the expansion of the market. Compliance with administrative guidelines ensures the safety and suitability of demonstrating devices, providing assurance to healthcare providers and patients. By assisting with information analysis, translation, and example recognition, simulated intelligence computations increase productivity and precision in demonstrations. As evidence ideal models are changing due to this mechanical coordination, which is also influencing market components. In spite of advancements, problems with the viability and accessibility of autoimmune disease diagnosis continue to exist particularly in developing regions. It is imperative that attempts to address this problem through important campaigns and teamwork be made in order to ensure that everyone in the globe has access to fair medical care. The global market for autoimmune illness diagnostics is poised for growth, propelled by ongoing research, technological developments, and an increasing understanding of autoimmune issues. Future components of the market will consist of a combination of rational and cooperative endeavours inside the medical care industry.



















Leave a Comment