Market Trends
Key Emerging Trends in the Atorvastatin API Market
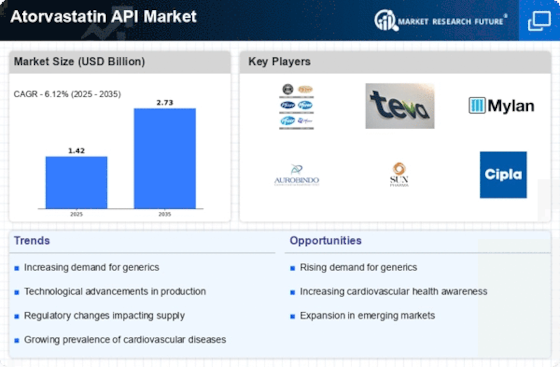
The market for a medicine called atorvastatin, which is used to treat heart-related problems, is expected to grow steadily at a rate of 3.34%. This medicine is widely used to treat cardiovascular diseases (CVDs), and there are opportunities for growth, especially in China, where more people are facing issues like high cholesterol. However, there are some health problems associated with this medicine that might slow down its growth globally.
Cardiovascular diseases (CVDs) are problems related to the heart and blood vessels, like heart disease and stroke. They are a leading cause of death worldwide, with over 18 million deaths in 2017. These diseases are often caused by high levels of glucose, blood pressure, and cholesterol, as well as being overweight. High cholesterol can block blood vessels, leading to chronic heart diseases. Atorvastatin is a medicine that helps regulate cholesterol levels in the body by reducing bad cholesterol and increasing good cholesterol. It is known to lower the risk of heart attacks and strokes in people with multiple risk factors like high blood pressure and diabetes.
One specific condition, hypercholesterolemia, is when there are high levels of cholesterol in the blood, leading to cardiovascular diseases, especially coronary heart disease. Atorvastatin works by preventing the formation of cholesterol in the liver.
Another condition, dyslipidemia, is when there are abnormal amounts of lipids (cholesterol and/or fat) in the blood, increasing the risk of coronary heart disease and stroke. Atorvastatin plays a crucial role in managing dyslipidemia by reducing the formation of cholesterol in the liver, similar to how it works for hypercholesterolemia. The medicine is available in different strengths and is expected to grow in demand due to rising concerns about cardiovascular diseases and the increased use of atorvastatin to treat these conditions.
In simpler terms, atorvastatin is a medicine that helps treat problems related to the heart and blood vessels. It's commonly used to manage conditions like high cholesterol, which, if left untreated, can lead to serious heart diseases. Atorvastatin works by regulating cholesterol levels in the body, reducing the bad cholesterol and increasing the good cholesterol. It's particularly useful in preventing heart attacks and strokes, especially in people with other risk factors like high blood pressure and diabetes. This medicine is crucial in addressing two specific conditions, hypercholesterolemia (high cholesterol levels in the blood) and dyslipidemia (abnormal amounts of lipids in the blood), both of which significantly increase the risk of heart-related issues. As more people become concerned about their heart health, the demand for atorvastatin is expected to grow, making it an essential player in the treatment of cardiovascular diseases globally.



















Leave a Comment