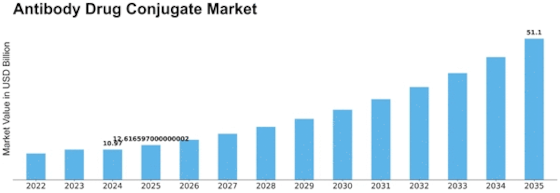
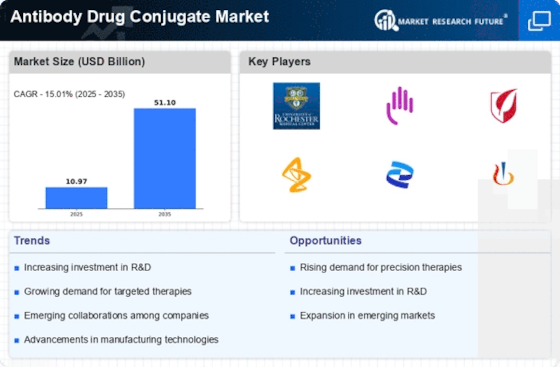
Antibody Drug Conjugate Size
Antibody Drug Conjugate Market Growth Projections and Opportunities
The scene of the antibody drug conjugate (ADC) market is unpredictably molded by a huge number of impacting factors. Relating to this particular area of the drug business, partners craving to gain by the valuable open doors should have a nuanced cognizance of these market factors.
Accuracy Medication and Designated Disease Treatments: The rising spotlight on designated malignant growth treatments and accuracy medication has considerably moved the ADC market. The nonstop movement of information in regard to disease science expands the requirement for treatment choices that are both exact and strong. ADCs, by prudence of their ability to regulate cytotoxic mixtures explicitly to malignant growth cells, are as per the precepts of accuracy medication.
Mechanical Progressions in Counter acting agent Designing: The ADC market is fundamentally affected by innovative advances in neutralizer designing. Progressions in payload conveyance frameworks, neutralizer plan, and formation strategies expand the particularity and adequacy of ADCs. Associations that designate assets towards state-of-the-art innovations accomplish an upper hand by creating prevalent items that address the consistently changing requests of the restorative business.
Oncology Medication Improvement Scene: The ADC market is significantly influenced by the more extensive scene of oncology drug advancement. The interest in ADCs is affected by the pervasiveness of specific disease types, the advancement of novel biomarkers, and the development of treatment standards. Organizations that better adjust their item improvement systems to the consistently changing oncology climate have a more prominent possibility of making progress.
Administrative Climate and Endorsements: The ADC market is fundamentally impacted by the administrative climate, explicitly the endorsement processes for novel therapeutics. Albeit thorough administrative norms ensure the security and viability of an item, they likewise present impediments as time and assets expected to enter the market. Associations that have the capacity to really explore administrative cycles can speed up the endorsement of their items, in this manner achieving an upper hand.
The ADC market is set apart by an exceptionally serious climate, which requires the execution of separation techniques. Associations that give particular ADC details, upgraded linker advances, or further developed security profiles separate themselves. As well as drawing in medical care experts, compelling separation impacts recommending designs, which adds to the development of piece of the pie.
The openness and reasonableness of ADC treatments for patients is a basic perspective that is influenced by different elements, including repayment strategies and the ability to pay. Associations whose evaluating methodologies work out some kind of harmony among maintainability and availability advance their market situating. Accomplishing broad patient access through medical services framework route is crucial for long haul market achievement.
Clinical Preliminary Outcomes and Genuine Proof: The validity and agreeableness of ADCs are impacted by the results of clinical preliminaries and the age of certifiable proof. Associations that allot assets towards complete clinical preliminary drives and reliably track experimental information can outfit medical care experts with convincing validation in regard to the adequacy and security of their items, consequently strengthening market trust.
Encouraging joint efforts and venturing into global business sectors are basic key targets for associations working in the ADC area. Possibilities for development are given by the cooperation of global accomplices and the variety of territorial medical care conditions. Associations that successfully oversee mind boggling worldwide administrative scenes and develop productive coalitions lay out an establishment for an extended market impression.
Patient and Doctor Training Drives: The ADC market is altogether affected by instructive drives that target the two patients and doctors. Associations that effectively partake in instructive missions improve public awareness as well as enable medical care specialists and patients to settle on very much educated choices. Upgrading cognizance helpfully affects market interest.



















Leave a Comment