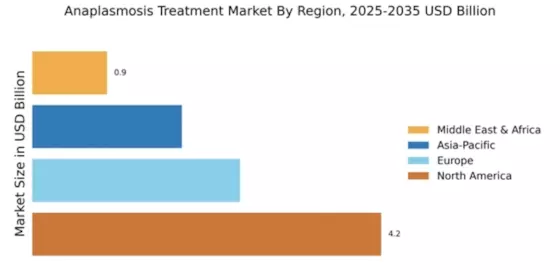
Market Growth Projections
The Global Anaplasmosis Treatment Market Industry is projected to experience substantial growth in the coming years. With a market value expected to reach 8.29 USD Billion in 2024, the industry is poised for significant expansion. The anticipated compound annual growth rate (CAGR) of 6.75% from 2025 to 2035 indicates a robust demand for treatment options as awareness, diagnostic capabilities, and healthcare infrastructure improve. This growth trajectory reflects the increasing recognition of anaplasmosis as a critical public health issue, prompting stakeholders to invest in research, development, and distribution of effective treatments.
Growing Awareness and Education
There is a notable increase in awareness and education regarding anaplasmosis, which is positively impacting the Global Anaplasmosis Treatment Market Industry. Public health campaigns and educational initiatives by health organizations are informing both healthcare professionals and the general public about the risks associated with tick-borne diseases. This heightened awareness leads to earlier diagnosis and treatment, ultimately improving patient outcomes. As more individuals recognize the symptoms and seek medical attention, the demand for effective treatment options is likely to grow, contributing to the projected market expansion to 17.0 USD Billion by 2035.
Rising Incidence of Anaplasmosis
The Global Anaplasmosis Treatment Market Industry is experiencing growth due to the increasing incidence of anaplasmosis, particularly in regions where tick populations are rising. The Centers for Disease Control and Prevention indicates that cases of anaplasmosis have been on the rise, with thousands of reported cases annually in the United States alone. This trend is likely to continue as climate change alters tick habitats, leading to more human exposure. As awareness of this disease grows, healthcare providers are more inclined to seek effective treatment options, thereby driving demand in the Global Anaplasmosis Treatment Market Industry.
Development of Targeted Therapies
The development of targeted therapies for anaplasmosis is emerging as a key factor in the Global Anaplasmosis Treatment Market Industry. Pharmaceutical companies are focusing on creating more effective treatments that specifically target the pathogens responsible for the disease. These therapies promise to enhance treatment efficacy and reduce side effects, making them more appealing to both healthcare providers and patients. As research progresses and new therapies enter the market, the demand for innovative treatment options is expected to rise, further driving market growth. This trend aligns with the overall expansion of the market, which is projected to reach 17.0 USD Billion by 2035.
Advancements in Diagnostic Techniques
Innovations in diagnostic techniques are significantly influencing the Global Anaplasmosis Treatment Market Industry. Enhanced testing methods, such as polymerase chain reaction (PCR) and serological assays, allow for quicker and more accurate diagnosis of anaplasmosis. These advancements not only facilitate timely treatment but also improve patient outcomes. As healthcare facilities adopt these technologies, the demand for effective treatment options is expected to rise. This shift is anticipated to contribute to the market's growth, with projections indicating a market value of 8.29 USD Billion in 2024, reflecting the increasing reliance on advanced diagnostics in managing anaplasmosis.
Increased Investment in Healthcare Infrastructure
Investment in healthcare infrastructure is a critical driver for the Global Anaplasmosis Treatment Market Industry. Governments and private entities are increasingly allocating resources to enhance healthcare facilities, particularly in endemic regions. This investment not only improves access to healthcare services but also facilitates the availability of advanced treatment options for anaplasmosis. Enhanced infrastructure allows for better disease surveillance and management, which is essential in controlling outbreaks. As healthcare systems strengthen, the market is expected to grow at a CAGR of 6.75% from 2025 to 2035, reflecting the positive impact of these investments on disease management.