Market Analysis
In-depth Analysis of Advance Anesthesia Monitoring Devices Market Industry Landscape
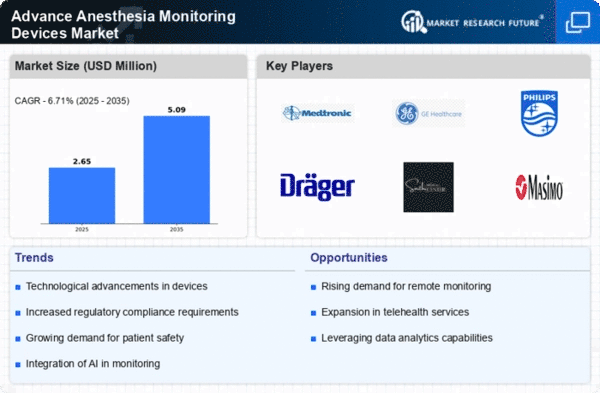
The market elements of Advance Anesthesia Monitoring Devices are driven by the rising interest for advanced innovations in medical care. These devices assume a pivotal part in guaranteeing patient wellbeing during surgeries by ceaselessly monitoring essential signs and anesthesia levels. The market is emphatically affected by the rising worldwide medical care use. As nations put more in further developing medical care foundation, there is an equal expansion in the reception of advanced clinical devices, including anesthesia monitoring devices. The surge in the quantity of surgeries universally is a vital driver for the market. Advance Anesthesia Monitoring Devices are fundamental in different restrained settings, going from routine medical procedures to complex methods, enhancing their interest across the medical care range. Developing awareness about patient security among medical services experts and patients has prompted an expanded accentuation on the utilization of monitoring devices. Anesthesia monitoring devices contribute essentially to lessening the dangers related with anesthesia organization, in this manner encouraging their market growth. The ubiquity of ongoing illnesses requires continuous clinical intercessions and medical procedures. This, thus, supports the interest for anesthesia monitoring devices as an imperative part in overseeing patients with constant medical issue through surgeries. The market is seeing a shift towards non-invasive monitoring techniques. Non-invasive anesthesia monitoring devices, which dispense with the requirement for invasive techniques, are getting some decent momentum because of their capacity to diminish patient inconvenience and improve medical care encounters. The globalization of medical care administrations has prompted a more interconnected existence where clinical prescribed procedures and innovations are shared across borders. This decidedly affects the reception of advanced clinical devices, including anesthesia monitoring devices, in various districts.



















Leave a Comment