Pune, India, June, 2022/MRFR Press Release/- Market research future has a half cooked research report on Europe Pediatric Neurology Device Market. The Europe market for pediatric neurology device is growing at a rapid pace and expected to reach a good market number by the end of the forecasted period 2024-2032.
Market Highlights
The Europe Pediatric Neurology Devices Market is expected to reach USD 156.34 Billion by 2032 at 6.04% CAGR during the forecast period 2024 -2032.The pediatric neurological device market is estimated to grow at a good pace and will achieve a significant growth in the market. The medical devices are being launched with a set of rules and regulations to provide safety, quality and a harmless product for the good public/patient health. The medical device industry is always under surveillance of rules and regulations and clinical trials. Also the number of audits are done several times. The medical device manufactures have to undergo all the quality test and have to follow the set of rules and regulations.
They need to maintain the standards regarding the manufacturing of devices and especially for the neurological devices used for children. There are a number of pathways available to bring a medical device to market. The regulatory procedures for a specific device depends on the classification of the device. These procedures are designed by the FDA who is majorly contributing and supporting to launch the medical devices for the pediatric use. There are comparatively few class I (lowest risk) neurological devices. Most neurological devices are classified as class II (moderate risk) or class III (high risk) devices. Examples of Class I devices include ventricular needles and anvils used to form skull plates. Examples of Class II devices include neurostimulators, aneurysm clips, and blood clot retrievers.
Examples of Class III devices include deep brain stimulators and medical devices for the treatment of brain tumors. The class to which your device is assigned determines, among other things, the type of premarket submission necessary, if required, to legally market your device. The European Pediatric Neurology Society is the other organization aggressively working for the pediatric healthcare. Various training, clinical care and scientific research are being carried out by this organization. Also the European regulatory bodies are working for the development of technologies in the pediatric neurology. European Association of Neurological Societies, European Academy of Neurology are constantly working for the Neurological disorders and to treat them effectively.
Further, looking at the current trends of the people in these regions. The lifestyle and the habits are leading to more number neurological disorders in children. With increasing diseases prevalence and also geriatric population the demand of Pediatric Neurology devices is expected to be enormous. Thus, this will eventually will result in the increasing demand for devices and products to monitor and also for the replaceable products. Further Pediatric Neurology Device have captured good market share. Based on the historical trends and market scenario, Pediatric Neurology device is expected to be the fastest growing segment of this market during 2022-2030.
Taste the market data and market information presented through more than 50 market data tables and figures spread over 50 numbers of pages of the project report. Avail the in-depth table of content TOC & market synopsis on “Pediatric Neurology Device Market Research Report –Europe Forecast to 2030.
Europe Pediatric Neurology Device Market Players:
Some of the major players in this market are: Medtronic, Elana Inc. Courtagen, Ethicon, Neumors, Inova Health system, Philadelphia Pediatric Medical Device Consortium and others.
Europe Pediatric Neurological Device Regional Report Analysis
Depending on European region, Pediatric Neurology Device market is segmented into countries like: Germany, France, Italy, Spain, U.K and rest of Europe. European market is estimated to be growing market during the forecasted period 2017-2023. These market have are estimated to good growing due to the factors that are leading the market growth are rising birth rate and the increasing prevalence of the neurological disorders. Also the European organizations are adopting new techniques for the pediatric care and the funds are also raised by the European organization for using new technology medical equipment.
Segmentation:
Europe Pediatric Neurology Device market has been segmented on the basis of services and treatment, neurological subspecialties and age groups. These further includes by Services & Treatment -Electroencephalogram, Intrathecal baclofen therapy, Lumbar puncture, Neurological evaluation, Vagal nerve Stimulation, by Neurological Subspecialties -Neuro-oncology, Neuromuscular, Neonatal neurology, Neuro-immunology, stroke and others, by Age Group- Neonates, Infants, Children, Adolescents.
Figure: Europe Pediatric Neurology Device Market Share by Neurological Subspecialties, 2016 (%)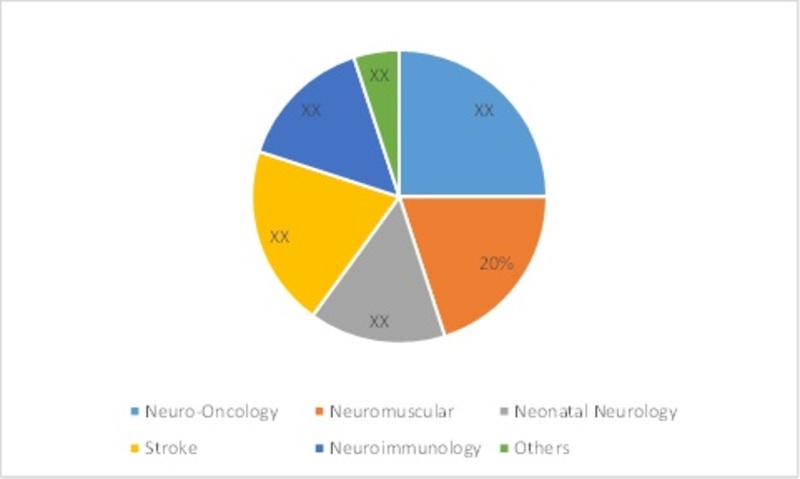
Browse In-depth Market Details [Table of Content, List of Figures, List of Tables] of Europe Pediatric Neurology Devices Market Research Report