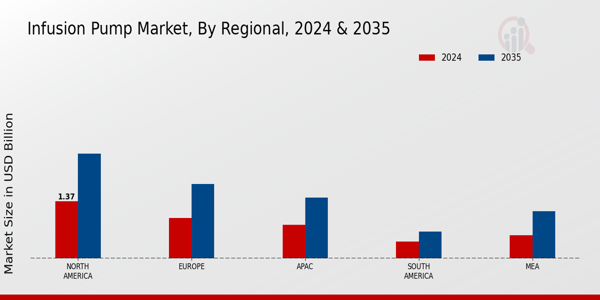
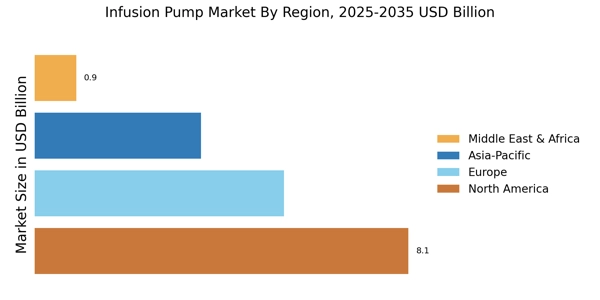
The secondary research process involved comprehensive analysis of regulatory medical device databases, peer-reviewed clinical engineering journals, healthcare technology publications, and authoritative medical organizations. Key sources included the US Food & Drug Administration (FDA) Center for Devices and Radiological Health (CDRH), FDA MAUDE (Manufacturer and User Facility Device Experience) recall database, European Medicines Agency (EMA), EUDAMED (European Database on Medical Devices), World Health Organization (WHO) Global Health Observatory, Organisation for Economic Co-operation and Development (OECD) Health Statistics, Centers for Medicare & Medicaid Services (CMS) Hospital Outpatient Quality Reporting data, Centers for Disease Control and Prevention (CDC) National Health Statistics Reports, National Institutes of Health (NIH) National Library of Medicine, National Center for Biotechnology Information (NCBI/PubMed) clinical infusion studies, American Society of Health-System Pharmacists (ASHP), Infusion Nurses Society (INS), European Society of Clinical Pharmacy (ESCP), American Association of Diabetes Educators, Oncology Nursing Society (ONS), American Society of Clinical Oncology (ASCO), European Society for Medical Oncology (ESMO), UN Comtrade (HS Code 901890 medical device trade flows), national medical device registries from key markets, and hospital procurement tender databases.
Volumetric pumps, syringe pumps, ambulatory pumps, insulin pumps, PCA pumps, enteral pumps, infusion accessories, adverse event reports, hospital bed-to-pump ratios, clinical safety studies, chronic disease prevalence data, procedure volume metrics, and competitive technology landscape analysis were all gathered from these sources.