Zika Virus Testing Market Overview
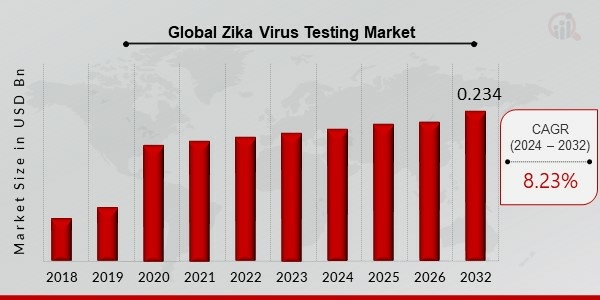
Zika Virus Testing Market Size was valued at USD 0.132 billion in 2023. The Zika Virus Testing Market industry is projected to grow from USD 0.145 billion in 2024 to USD 0.234 billion by 2032, exhibiting a compound annual growth rate (CAGR) of 8.23% during the forecast period (2024 - 2032).Infection with the Zika virus is spread via mosquito bites. As a result, Zika is a mosquito-borne illness spread by the Aedes genus. Zika is spread by mosquitos, like other mosquito-borne diseases, and can be passed from one person to another through sexual contact.
A pregnant woman who is infected with the virus may pass it on to her unborn child.
Increasing efforts by the US FDA (United States Food and Drug Administration) to restrict the transmission of this virus are expected to boost the Zika Virus Testing Market from 2024 to 2032. (Forecast period). Furthermore, the US Department of Health and Human Services has taken steps to prevent the virus from infecting other individuals. These measures have also increased the demand for Zika virus testing in the United States and other parts of the world. The market will soon see huge growth as per the forecasts.
Mylab, in July 2022, launched a Zika virus Real Time-PCR test kit. The two-hour duration within which the combined kit produces results has made it suitable for mass testing.For example, in May 2022, the Uttar Pradesh Government (India) launched ‘Zero Mission’ ‘Zero’ to eradicate vector-borne diseases such as Japanese encephalitis, dengue fever, malaria, typhoid fever, pneumonia and Zika virus.
This is the primary reason behind the growing prevalence of Zika virus infection across the world and initiatives by governments during the forecast period.In March 2022, for instance, an international team of researchers led by University of Toronto experts presented early results from one of the first field trials demonstrating synthetic biology-based diagnostics using real patient samples.
Conducted on-site in Latin America, these experiments show how cell-free synthetic biology tools and portable hardware can be employed to produce immediate, decentralized, low-cost patient testing for tropical infectious diseases like the Zika virus.The World Health Organization (WHO) established its program regarding emerging and re-emerging arbovirus infections such as Chikungunya fever, Dengue fever, Yellow Fever and Zika Virus on March 31st, 2017. Tropical and subtropical regions are now under the threat of Aborvirus infection due to an increased frequency of incidence.
Market growth will be influenced by several factors such as enhanced research activities, development of Zika vaccines and high health care spending. Indian Council for Medical Research (ICMR), together with Klenzaids, jointly established India’s first Mobile BSL-3 enhanced laboratory in Maharashtra, India, in February 2022. Costing over INR 25 crores, it was built at a cost of about INR Crore-Klenzaids said on their website that “The entire stand-alone operation by any means includes each system & equipment required for BSL-3 Enhanced laboratory.”
Zika Virus Testing Market Segment Insights
The global industry is split on account of tests, end-users, and regions.
Zika Virus Testing Test Insights
The tests include serological/Zika virus antibody and molecular/nucleic acid amplification.
Zika Virus Testing End-User Insights
Diagnostic units, hospitals, pathology labs, etc. form the end-users.
Zika Virus Testing Regional Insights
By regions, the Zika virus testing market is divided on the basis of Asia Pacific, Europe, Americas, and the Middle East and Africa.
The constant transmission of Zika in various American nations is reported to augment the need for testing services till 2030. Moreover, projects undertaken by the HHS, BARDA to boost diagnostic assay innovations for the identification and treatment of this illness could propel industry expansion over the forecast period.
Additionally, there is more outburst of the Zika virus across multiple Latin American nations. For instance, twenty one of the twenty six states in Brazil are affected by the Zika virus. Faster transmission in the world is projected to lead to the creation of more detection assays, thus steering the market incomes.
Recent Development
The FDA has given the commercial Altona Diagnostics RealStar ZIKV RT-PCR test kit (Altona PCR) emergency use authorization. The goal of this study was to compare the Altona PCR to the CDC-designed dual-target ZIKV RT-PCR reference assay (reference PCR) and to characterize the demographics of ZIKV RT-PCR patients in Ontario, Canada. The Altona PCR and the reference PCR were used to test a significant number of clinical specimens for ZIKV. Positive and ambiguous specimens were subjected to ZIKV NS5 gene PCR and Sanger sequencing.
Luminex Corporation is a biotechnology firm that creates, produces, and distributes patented biological diagnostic methods and products. Flow cytometers, cellular analysis devices, a platform for monitoring genes and proteins, and other diagnostics and research tools are among the company's products. Clinical diagnostics, pharmaceutical drug development, biomedical research, genomic and proteomic research, and food safety are among the markets served by Luminex.
Eminent Players and Industry Advancements
Prominent companies affiliated with the Zika virus testing market comprise Luminex Corporation, ARUP Laboratories, Roche Molecular Systems, Hologic, and Siemens Healthcare Diagnostics. To create efficient treatments and vaccines, advanced animal models are required. An improved comprehension of immunological protection correlates is also needed to protect ourselves from this disease.
Nowadays, the Zika virus is detected in view of clinical manifestations. Molecular detection and serological devices also play a major role. As the separation of this virus is difficult; LFAs, NASBA, and LAMP are used to detect the virus and ascertain the cause of the fever. These novel techniques have assisted in accurate & fast diagnosis. With the clinical symptoms of Zika not being pathognomic, the presence of this virus can be established only when it is separated and examined.
Few among many clinical symptoms of this disease are myalgia, headache, fever, maculopapular rashes, and arthralgia. Only upon further diagnosis and confirmation, are the treatments recommended to patients. Sometimes, similar symptoms can be mistaken for other illnesses. Thus, it’s best to go to the doctors and have it ruled in/out.
Key Players:
Zika Virus Testing Market Industry Developments
-
Q2 2024: FDA Authorizes Emergency Use of New Zika Virus RT-PCR Test Developed by BioGenDx The FDA granted Emergency Use Authorization to BioGenDx for its new rapid RT-PCR test for Zika virus, enabling faster detection during ongoing outbreaks in the Americas.
-
Q1 2024: Roche Launches Next-Generation Zika Virus Diagnostic Test in Latin America Roche announced the commercial launch of its next-generation Zika virus molecular diagnostic test, designed for high-throughput laboratories, in Brazil and other Latin American countries.
-
Q3 2024: Siemens Healthineers Partners with Brazilian Ministry of Health for Zika Virus Testing Rollout Siemens Healthineers signed a partnership agreement with Brazil's Ministry of Health to supply advanced Zika virus testing platforms to public health laboratories amid rising case numbers.
-
Q2 2024: Thermo Fisher Scientific Expands Zika Virus Test Production Capacity in Singapore Facility Thermo Fisher Scientific announced the expansion of its Singapore manufacturing facility to increase production of Zika virus molecular diagnostic kits, responding to heightened demand in Southeast Asia.
-
Q4 2024: Abbott Receives CE Mark for Multiplex Zika, Dengue, and Chikungunya Test Abbott Laboratories received CE Mark approval for its multiplex molecular assay that simultaneously detects Zika, dengue, and chikungunya viruses, enabling broader deployment in European and global markets.
-
Q1 2025: QuidelOrtho Acquires Zika Rapid Test Startup ViraSure for $120 Million QuidelOrtho completed the acquisition of ViraSure, a startup specializing in rapid Zika virus diagnostic tests, to strengthen its infectious disease testing portfolio.
-
Q2 2025: Bio-Rad Laboratories Launches Point-of-Care Zika Virus Antibody Test in Africa Bio-Rad Laboratories launched a new point-of-care Zika virus IgM/IgG antibody test, targeting rapid diagnosis in remote and resource-limited African regions.
-
Q3 2024: GenMark Diagnostics Announces $50 Million Funding Round to Accelerate Zika Test Development GenMark Diagnostics disclosed in an SEC filing that it raised $50 million in new funding to accelerate the development and regulatory submission of its next-generation Zika virus diagnostic platform.
-
Q2 2024: Hologic Wins Major Zika Virus Testing Contract from Pan American Health Organization Hologic secured a multi-year contract with the Pan American Health Organization to supply Zika virus molecular testing kits to public health labs across Central and South America.
-
Q4 2024: CDC Updates Zika Virus Testing Guidelines and Approves New Diagnostic Panel The U.S. Centers for Disease Control and Prevention (CDC) updated its Zika virus testing guidelines and approved a new diagnostic panel for use in state and local public health laboratories.
-
Q1 2025: Siemens Healthineers Appoints New Head of Zika Virus Diagnostics Division Siemens Healthineers announced the appointment of Dr. Maria Alvarez as the new head of its Zika virus diagnostics division, aiming to drive innovation and global market expansion.
-
Q2 2025: Roche Receives ANVISA Approval for Zika Virus PCR Test in Brazil Roche received regulatory approval from Brazil's ANVISA for its Zika virus PCR test, enabling broader clinical use in the country amid ongoing public health concerns.
| Report Attribute/Metric |
Details |
| Market Size 2023 |
0.132 (USD Billion) |
| Market Size 2024 |
0.145 (USD Billion) |
| Market Size 2032 |
0.234 (USD Billion) |
| Compound Annual Growth Rate (CAGR) |
8.23 % (2024 - 2032) |
| Report Coverage |
Revenue Forecast, Competitive Landscape, Growth Factors, and Trends |
| Base Year |
2023 |
| Market Forecast Period |
2024 - 2032 |
| Historical Data |
2019 - 2023 |
| Geographies Covered |
North America, Europe, Asia-Pacific, and Rest of the World (RoW) |
| Key Vendors |
ARUP Laboratories, Hologic, Luminex Corporation, Roche Molecular Systems, etc. |
| Key Market Opportunities |
The increasing number of Zika virus cases globally creates a significant demand for effective testing solutions. |
| Key Market Drivers |
The increasing number of Zika virus cases globally, especially in tropical and subtropical regions. |
Zika Virus Testing Market Highlights:
















