North America : Leading Market for Innovation
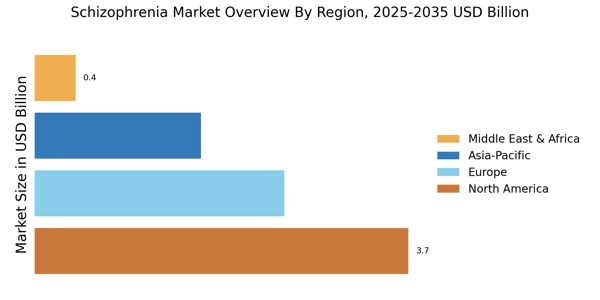
North America leads in the Schizophrenia Market size, accounting for over 45% of the global revenue in 2024. The region benefits from advanced healthcare infrastructure, high R&D investments, and a growing prevalence of mental health disorders. Regulatory support from agencies like the FDA accelerates the approval of new therapies, driving market growth.
- CDC estimates 20.6% of U.S. adults (over 52 million people) experience mental illness annually, while federal mental health funding exceeds USD 4 billion per year, significantly supporting innovation, early diagnosis, and access to advanced schizophrenia treatments.
The increasing awareness of mental health issues further fuels demand for effective treatments. The United States is the primary contributor to this market, with significant presence from key players such as Johnson & Johnson, Eli Lilly, and Pfizer. The competitive landscape is characterized by a mix of established pharmaceutical giants and emerging biotech firms. The focus on innovative therapies, including antipsychotics and adjunctive treatments, positions North America as a hub for schizophrenia management and research.
Europe : Emerging Market with Growth Potential
Europe Schizophrenia Market size was valued at USD 2.49 billion in 2024, making it the second-largest regional market with a 30% share. Factors such as increasing mental health awareness, supportive healthcare policies, and a rise in diagnosed cases are driving this growth. The European Medicines Agency (EMA) plays a crucial role in regulating and approving new therapies, which enhances market dynamics and encourages innovation in treatment options.
- WHO Europe reports over 110 million people live with mental health conditions, representing nearly 1 in 9 individuals, with some countries allocating 8–10% of total health budgets to mental healthcare, driving improved diagnosis and treatment accessibility.
Leading countries in this region include Germany, France, and the UK, where significant investments in mental health services are being made. The competitive landscape features major players like Roche and AstraZeneca, alongside local firms focusing on tailored therapies. The emphasis on personalized medicine and integrated care models is shaping the future of schizophrenia treatment in Europe.
Asia-Pacific : Rapidly Growing Market Segment
The Asia-Pacific region is emerging as a significant player in the schizophrenia market, accounting for about 20% of the global share. The growth is driven by increasing awareness of mental health issues, rising disposable incomes, and expanding healthcare access. Countries like Japan and China are leading this growth, supported by government initiatives aimed at improving mental health care and reducing stigma associated with mental disorders.
Japan is a key market, with major pharmaceutical companies like Otsuka Pharmaceutical and Novartis actively involved in developing innovative treatments. The competitive landscape is evolving, with both multinational corporations and local firms striving to meet the growing demand for effective schizophrenia therapies. The focus on research and development is expected to enhance treatment options in the region.
Middle East and Africa : Untapped Market with Challenges
The Middle East and Africa region represents an untapped market for schizophrenia treatments, holding approximately 5% of the global market share. The growth is hindered by challenges such as limited healthcare infrastructure, stigma surrounding mental health, and varying levels of access to treatment. However, increasing awareness and government initiatives are beginning to address these issues, creating opportunities for market expansion.
Countries like South Africa and the UAE are making strides in mental health care, with efforts to improve access to treatment and support services. The competitive landscape is still developing, with a mix of local and international players. The presence of key pharmaceutical companies is gradually increasing, focusing on affordable and effective treatment options to cater to the unique needs of the region.