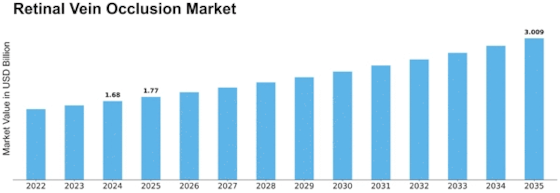
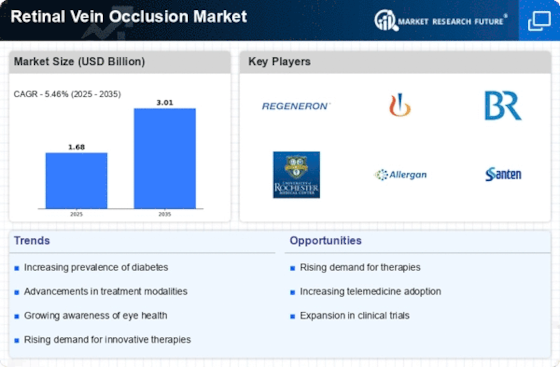
Retinal Vein Occlusion Size
Retinal Vein Occlusion Market Growth Projections and Opportunities
Retinal vein occlusion (RVO) is a common eye problem that can lead to vision loss, making it one of the most widespread causes of vision problems worldwide. Even though it's the second most common reason for vision loss due to retinal vascular disease, it still affects a lot of people. The market for dealing with retinal vein occlusion is expected to grow a lot because more people are getting eye diseases and other chronic conditions like diabetes, which can also harm the eyes. The demand for better treatments for eye problems is increasing, giving a boost to the market. Many companies in this market have opportunities to grow by using smart plans like expanding to different places. According to the World Health Organization, about 60 million people in Europe have diabetes, and diabetes is a big risk for eye diseases. In the United States, nearly 14 million people aged 12 and older have vision problems. In China, more people are getting diabetic retinopathy, especially with the rise in diabetes cases. Japan has 7.2 million people with diabetes, and this number is going up because of lifestyle changes and an increase in diseases like obesity. In Australia, almost 1.2 million people, which is about 5.1% of the population, have been diagnosed with diabetes.
The reason the market for treating retinal vein occlusion is growing is because more and more people are facing eye problems, especially due to conditions like diabetes. Diabetes is a significant risk factor for eye diseases, and as the number of people with diabetes increases, so does the need for effective treatments for eye issues like retinal vein occlusion. The demand for better and more advanced methods to treat these eye problems is driving the growth of the market. Companies that offer solutions for retinal vein occlusion have a great chance to expand their business, especially by reaching more places around the world. This is important because eye diseases and conditions are becoming more common globally.
Looking at the numbers, it's clear that diabetes is a big player in the rise of eye problems. In Europe, a massive 60 million people are dealing with diabetes, putting them at risk for eye diseases. In the United States, around 14 million people, who are 12 years and older, are struggling with vision issues. China is seeing an increase in diabetic retinopathy as the number of people with diabetes rises. In Japan, where 7.2 million people have diabetes, the cases are going up due to lifestyle changes and the growth of diseases like obesity. Australia has almost 1.2 million people diagnosed with diabetes, making up about 5.1% of the population. All these numbers point to a growing need for effective treatments for eye problems, pushing the retinal vein occlusion market to expand.



















Leave a Comment