Real World Evidence Market Summary
According to Market Research Future analysis, the Real-World Evidence Market size was valued at USD 3.54 Billion in 2024. The market is projected to grow from USD 4.15 Billion in 2025 to USD 20.3 Billion by 2035, exhibiting a CAGR of 17.2% during the forecast period 2025–2035. North America led the market with over 59.29% share, generating around USD 0.7 billion in revenue.
The Real-World Evidence Market is primarily driven by growing demand for personalized medicine and rising healthcare costs, encouraging stakeholders to leverage real-world data for treatment effectiveness, regulatory compliance, and value-based care decisions, ultimately improving patient outcomes and accelerating evidence-based healthcare innovation globally.
According to CDC, 95% of U.S. office-based physicians adopted electronic health records in 2024, significantly expanding real-world data generation. Additionally, WHO highlights digital health as critical for improving health system efficiency and evidence-based decision-making, supporting broader Real-World Evidence adoption.
Key Market Trends & Highlights
The Real-World Evidence Market is experiencing robust growth driven by technological advancements and regulatory support.
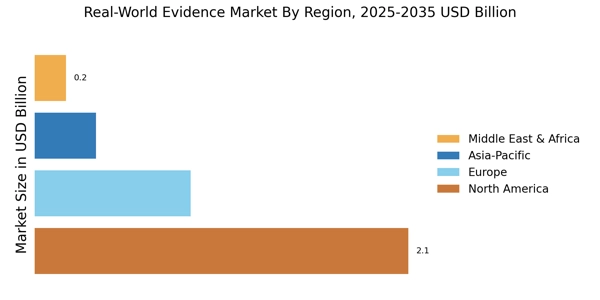
- North America dominated market with over 59.29% revenue share in 2024.
- Europe valued at USD 1.0 billion in 2024, projected to reach USD 3.2 billion by 2035.
- Drug Development segment accounted for approximately 41% share due to growing pharmaceutical R&D investments.
- Electronic Health Records contributed nearly 39% share driven by expanding healthcare digitalization globally.
Market Size & Forecast
| 2024 Market Size | 3.542 (USD Billion) |
| 2035 Market Size | 20.3 (USD Billion) |
| CAGR (2025 - 2035) | 17.2% |
Major Players
Companies such as Optum (US), IBM Watson Health (US), Celerion (US), Medidata Solutions (US), Parexel International(US), Roche (CH), Aetion (US), Flatiron Health (US), Syapse (US), Pharmerit International (US) are some of the major participants in the global market.