

- Global Market Outlook
- In-depth analysis of global and regional trends
- Analyze and identify the major players in the market, their market share, key developments, etc.
- To understand the capability of the major players based on products offered, financials, and strategies.
- Identify disrupting products, companies, and trends.
- To identify opportunities in the market.
- Analyze the key challenges in the market.
- Analyze the regional penetration of players, products, and services in the market.
- Comparison of major players’ financial performance.
- Evaluate strategies adopted by major players.
- Recommendations
- Vigorous research methodologies for specific market.
- Knowledge partners across the globe
- Large network of partner consultants.
- Ever-increasing/ Escalating data base with quarterly monitoring of various markets
- Trusted by fortune 500 companies/startups/ universities/organizations
- Large database of 5000+ markets reports.
- Effective and prompt pre- and post-sales support.
Market Size Snapshot
| Year | Value |
|---|---|
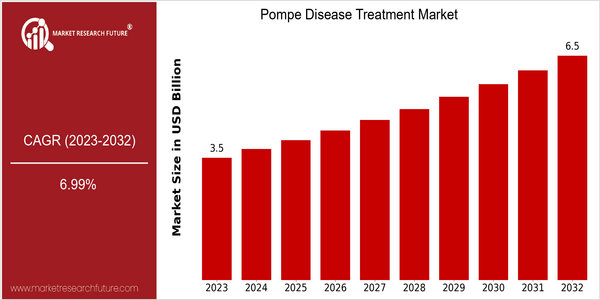
| 2023 | USD 3.54 Billion |
| 2032 | USD 6.5 Billion |
| CAGR (2024-2032) | 6.99 % |
Note – Market size depicts the revenue generated over the financial year
The Pompe Disease Treatment Market is currently valued at approximately USD 3.54 billion in 2023 and is projected to reach USD 6.5 billion by 2032, reflecting a robust compound annual growth rate (CAGR) of 6.99% from 2024 to 2032. This growth trajectory indicates a significant increase in demand for effective treatment options for Pompe disease, a rare genetic disorder characterized by the accumulation of glycogen in the body's cells due to enzyme deficiency. The rising prevalence of this condition, coupled with advancements in biotechnology and pharmaceuticals, is driving the market expansion. Several factors are contributing to the growth of the Pompe Disease Treatment Market. Key among them are the increasing investments in research and development aimed at innovative therapies, including enzyme replacement therapies and gene therapies. Companies such as Sanofi, which has made substantial investments in the development of its enzyme replacement therapy, and Amicus Therapeutics, known for its gene therapy initiatives, are at the forefront of this market. Strategic partnerships and collaborations between pharmaceutical companies and research institutions are also enhancing the pipeline of potential treatments, thereby fostering a competitive landscape that is likely to accelerate market growth in the coming years.
Regional Market Size
Regional Deep Dive
The Pompe Disease Treatment Market is characterized by a growing awareness of the disease and advancements in therapeutic options across various regions. In North America, the market is driven by a robust healthcare infrastructure, significant investment in research and development, and a high prevalence of genetic disorders. Europe showcases a strong regulatory framework and collaborative efforts among healthcare providers and pharmaceutical companies, while the Asia-Pacific region is witnessing an increase in healthcare access and investment in biotechnology. The Middle East and Africa face unique challenges, including limited healthcare resources, but are gradually improving through international partnerships. Latin America is experiencing a rise in awareness and diagnosis, leading to increased demand for treatment options. Overall, the market dynamics in each region reflect a combination of healthcare advancements, regulatory environments, and cultural factors that influence treatment accessibility and innovation.
Europe
- The European Medicines Agency (EMA) has streamlined the approval process for rare disease treatments, facilitating quicker access to therapies for Pompe disease patients.
- Collaborative initiatives, such as the European Reference Networks (ERNs), are enhancing knowledge sharing and treatment protocols among healthcare providers across Europe, which is expected to improve patient care.
Asia Pacific
- Countries like Japan and Australia are increasing their focus on rare diseases, with government programs aimed at subsidizing treatment costs and improving patient access to therapies.
- Biotechnology firms in China are emerging as key players in the Pompe disease treatment market, with several companies developing novel therapies that are expected to enter clinical trials soon.
Latin America
- Countries like Brazil and Argentina are implementing national health programs aimed at increasing awareness and diagnosis of rare diseases, including Pompe disease, which is expected to drive demand for treatment.
- Local pharmaceutical companies are beginning to collaborate with international firms to develop affordable treatment options tailored to the economic realities of the region.
North America
- The U.S. Food and Drug Administration (FDA) has recently approved new enzyme replacement therapies for Pompe disease, enhancing treatment options and accessibility for patients.
- Major pharmaceutical companies, such as Sanofi and Amicus Therapeutics, are investing heavily in clinical trials and research to develop innovative therapies, which is expected to significantly improve patient outcomes.
Middle East And Africa
- The World Health Organization (WHO) is working with local governments to improve genetic disorder awareness and screening programs, which is crucial for early diagnosis and treatment of Pompe disease.
- Partnerships between international NGOs and local healthcare providers are helping to improve access to treatment and education about Pompe disease in underserved regions.
Did You Know?
“Pompe disease affects approximately 1 in 40,000 live births, but its prevalence can be higher in certain populations, such as those of Dutch descent, where it occurs in about 1 in 3,200 births.” — National Institutes of Health (NIH)
Segmental Market Size
The Pompe Disease Treatment Market is characterized by a growing segment focused on enzyme replacement therapies (ERT) and gene therapies, which are crucial for managing this rare genetic disorder. The demand for these treatments is driven primarily by the increasing prevalence of Pompe disease, heightened awareness among healthcare providers, and advancements in biotechnology that facilitate the development of more effective therapies. Regulatory support, such as expedited approval processes for orphan drugs, further propels this segment's growth. Currently, the adoption stage for Pompe disease treatments is transitioning from pilot phases to more widespread deployment, with companies like Sanofi and Amicus Therapeutics leading the charge in innovative therapies. Primary applications include the administration of ERT to improve muscle function and quality of life for patients. Notable trends accelerating growth include the rise of personalized medicine and ongoing clinical trials exploring novel gene therapies. Technologies such as CRISPR and next-generation sequencing are shaping the evolution of treatment methodologies, enabling more targeted and effective interventions for patients suffering from Pompe disease.
Future Outlook
The Pompe Disease Treatment Market is poised for significant growth from 2023 to 2032, with an anticipated market value increase from $3.54 billion to $6.5 billion, reflecting a robust compound annual growth rate (CAGR) of 6.99%. This growth trajectory is underpinned by advancements in enzyme replacement therapies (ERTs) and gene therapies, which are expected to enhance treatment efficacy and patient outcomes. As awareness of Pompe disease increases and diagnostic capabilities improve, the market is likely to see a rise in patient identification and treatment initiation, potentially increasing penetration rates from current estimates of 20% to over 35% by 2032. Key technological drivers, including the development of next-generation ERTs and innovative gene-editing techniques, are set to revolutionize treatment paradigms. Additionally, supportive policy frameworks and increased funding for rare disease research will further catalyze market expansion. Emerging trends such as personalized medicine and patient-centric approaches are expected to shape treatment strategies, leading to more tailored therapies that address the unique needs of Pompe disease patients. As the market evolves, stakeholders must remain agile to capitalize on these trends and ensure sustainable growth in the coming decade.
Covered Aspects:| Report Attribute/Metric | Details |
|---|---|
| Market Size Value In 2022 | USD 1.1 Billion |
| Market Size Value In 2023 | USD 1.14 Billion |
| Growth Rate | 3.80% (2023-2032) |
Pompe Disease Treatment Market Highlights:
Leading companies partner with us for data-driven Insights
Kindly complete the form below to receive a free sample of this Report
Tailored for You
- Dedicated Research on any specifics segment or region.
- Focused Research on specific players in the market.
- Custom Report based only on your requirements.
- Flexibility to add or subtract any chapter in the study.
- Historic data from 2014 and forecasts outlook till 2040.
- Flexibility of providing data/insights in formats (PDF, PPT, Excel).
- Provide cross segmentation in applicable scenario/markets.









