Market Trends
Key Emerging Trends in the Paroxysmal Atrial Tachycardia Market
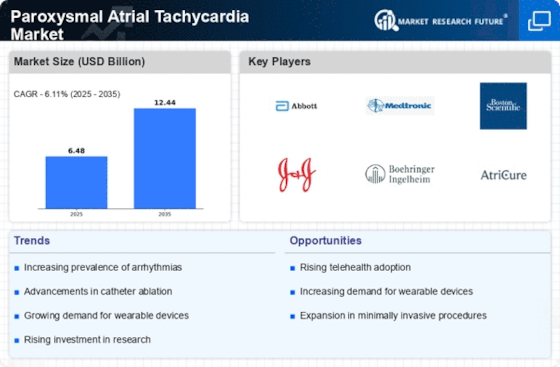
The market for paroxysmal atrial tachycardia (PAT), a form of arrhythmia characterized by fast heart rates originating within the atria, is witnessing a consistent boom in occurrence globally. Factors inclusive of older populations, growing prevalence of cardiovascular hazard elements, and stepped-forward detection techniques contribute to the developing burden of PAT. Catheter ablation has emerged as a general treatment modality for PAT patients refractory to pharmacological therapy or the ones looking for an extra definitive remedy choice. Radiofrequency or cryoablation strategies target particular areas of strange electrical activity inside the heart, efficaciously interrupting the circuit answerable for PAT and restoring normal coronary heart rhythm. Technological advancements in catheter ablation strategies and equipment contribute to improved procedural fulfillment costs and affect personal results in the PAT market. Advances, which include contact pressure sensing, three-dimensional mapping systems, and novel catheter designs, beautify procedural precision, efficacy, and safety. Non-invasive treatment options for PAT, which include vagal maneuvers, adenosine management, and electrical cardioversion, are powerful in terminating acute episodes of tachycardia and restoring sinus rhythm. These interventions are especially useful in the extreme control of PAT in emergency or outpatient settings. Remote monitoring answers play a vital role in the management of PAT sufferers, making an allowance for non-stop monitoring of coronary heart rhythm and well-timed detection of arrhythmia recurrences. Implantable cardiac devices, such as pacemakers and implantable cardioverter defibrillators (ICDs), with far-flung monitoring talents, allow healthcare companies to display PAT patients remotely and intervene promptly while essential. Patient education and lifestyle changes are critical additives of PAT control, empowering sufferers to understand triggers, adhere to treatment regimens, and undertake heart-healthy behaviors. Education on symptom popularity, medication adherence, nutritional adjustments, and pressure management enables PAT sufferers to manage their circumstances and improve universal consequences. Comorbidity control plays a big role in the complete care of PAT sufferers, given the association between PAT and underlying cardiovascular conditions, together with high blood pressure, coronary artery disorder, and heart failure. Optimal control of comorbidities through lifestyle adjustments and pharmacological interventions allows for lessening the danger of PAT recurrence and cardiovascular headaches. Ongoing research and improvement efforts are focused on raising awareness of novel healing targets and remedy modalities within the PAT market. Clinical trials investigating the efficacy and protection of emerging pharmacological agents, ablation techniques, and tool-primarily based treatment plans purpose to enlarge treatment alternatives and improve results for PAT patients. Intensifying competition amongst pharmaceutical agencies, scientific device producers, and healthcare companies influences market dynamics in the PAT market. Companies differentiate their products and services through technological improvements, medical efficacy, and pricing strategies to benefit an aggressive side and seize market percentage.


















Leave a Comment