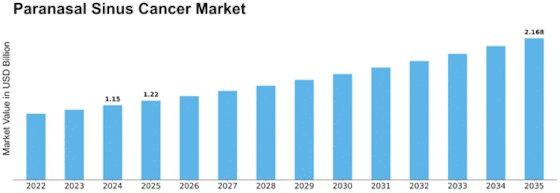
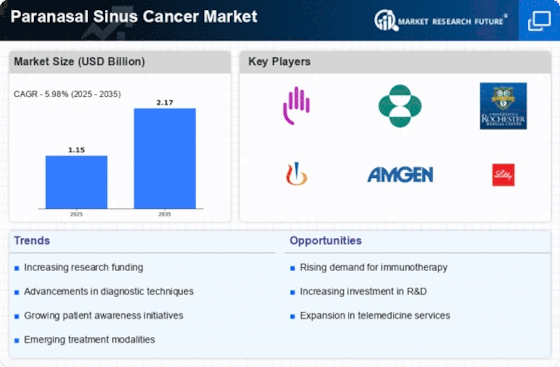
Paranasal Sinus Cancer Size
Paranasal Sinus Cancer Market Growth Projections and Opportunities
The frequency and severity of paranasal sinus carcinoma impact the therapy market. As more patients are recognized, they demand effective medications, changing the market. New imaging and biopsy technologies increase paranasal sinus carcinoma industry. Early and accurate diagnosis is crucial for treatment, and diagnostic advancements have improved patient outcomes. Research and development enhance paranasal sinus carcinoma therapy. Targeted and immunotherapies are innovative medications that improve patient choices and transform the market. The principles for treating paranasal sinus carcinoma are crucial. Market entry requires health authority approval. New treatments may take longer to reach patients if laws change or approval speeds up. Patient demographics, notably age distribution, impact the market. Paranasal sinus carcinoma is more common in those over 65, hence the therapy market may expand. Healthcare infrastructure benefits the market. New medications may be swiftly adopted in countries with excellent healthcare, but the market may not expand. Early detection and public awareness programs impact markets. If individuals recognize the symptoms and risk factors of paranasal sinus cancer, it may be identified sooner, changing treatment and market demand. Pharmaceutical, research, and healthcare alliances affect the market. Research, strategic alliances, and new medications may accelerate the process and influence market competition. Health insurance benefits and payment impact care access. Therapy coverage and reimbursement may determine how many patients obtain paranasal sinus cancer therapy.
GDP and health care expenditure impact the market. If the economy is steady, healthcare funding may increase, leading to greater research and cheaper treatment costs. Finding the genetic and environmental causes of paranasal sinus carcinoma might shift the market. Genetic research may lead to specialized medications, while external risk factors may safeguard and trend markets.



















Leave a Comment