Market Analysis
In-depth Analysis of Intracranial Hemorrhage Diagnosis and Treatment Market Industry Landscape
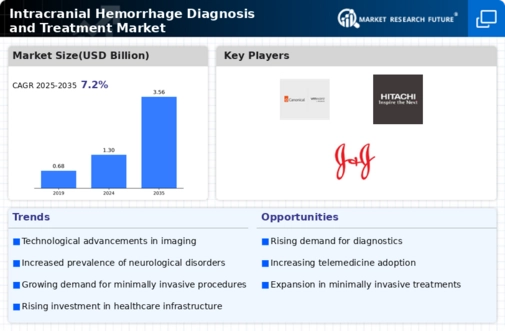
The world is witnessing a rise in traumatic incidents and accidents, leading to a condition known as Traumatic Brain Injury (TBI). According to the World Health Organization (WHO), these incidents have significant economic implications, costing most countries around 3% of their gross domestic product. Shockingly, road traffic injuries rank as the leading cause of death among children and young adults aged 5-29 years. Each year, approximately 1.35 million lives are lost due to road traffic crashes, with an additional 20 to 50 million people suffering non-fatal injuries. These injuries often result in disabilities, creating a lasting impact on the lives of those affected. The consequences extend beyond physical injuries, as damage to the brain can lead to persistent pain for many years following the accident. A 2018 article in the Journal of Neurosurgery estimated that a staggering 69 million individuals worldwide have experienced TBI from various causes. Regions such as Southeast Asia and the Western Pacific bear the greatest burden of this disease.
One of the primary contributors to the increasing prevalence of TBI is head injuries resulting from road traffic collisions, especially in low and middle-income countries. The proportion of TBIs attributed to road traffic collisions is significantly higher in these nations. This alarming trend of road accidents and subsequent TBIs is propelling the growth of the global intracranial hemorrhage diagnosis and treatment market.
Amidst these challenges, clinical trials are playing a crucial role in advancing medical knowledge and finding effective treatments. Clinical trials serve as a vital platform for discovering not only new treatments for diseases but also innovative methods of detection, diagnosis, and prevention. As of December 12, 2020, there were 125 ongoing phase 4 clinical trials assessing the efficacy of various drugs and procedures for treating intracranial hemorrhage. Companies actively involved in these trials include Portola Pharmaceuticals, Otsuka Pharmaceutical Co., Ltd, PharmEvo Pvt Ltd, Bayer AG, Boehringer Ingelheim, and Cumberland Pharmaceuticals.
Research funding is a key enabler of these clinical trials. The Canada Brain Research Fund, for example, allocated USD 230 million in new funding by June 2019 to support 300 projects across Canada, involving over 1,000 researchers and more than 115 institutions. This funding facilitates collaboration among research institutes, allowing them to conduct clinical trials and contribute to the advancement of medical knowledge. An example is Oslo University Hospital in Norway, which is currently evaluating the effects of antithrombotic drugs for preventing ischemic events in patients with recent intracerebral hemorrhage.
The availability of research funds and the substantial number of clinical trials in the fourth phase are expected to drive progress in the intracranial hemorrhage diagnosis and treatment market. These efforts hold the promise of introducing novel drugs and devices, ultimately improving the prognosis and quality of life for individuals affected by traumatic incidents and TBIs.


















Leave a Comment