Research Methodology on Heart Failure Drugs Market
1. Introduction
This research report focuses on the market analysis of heart failure drugs. The main objective of this report is to provide insights into the market size and trends of the heart failure drugs market. This study includes data collected from primary and secondary sources. The primary data includes interviews with individuals working in the heart failure drugs market, industry participants, stakeholders, and the secondary data includes periodicals, government publications, investor presentations, third-party databases, and published sources for the heart failure drugs market.
2. Research Scope
The scope of this research report is to provide comprehensive information on the global heart failure drugs market. The report provides an in-depth analysis of the market size and trends of the heart failure drugs market. This report analyzes the market according to type, application and geography.
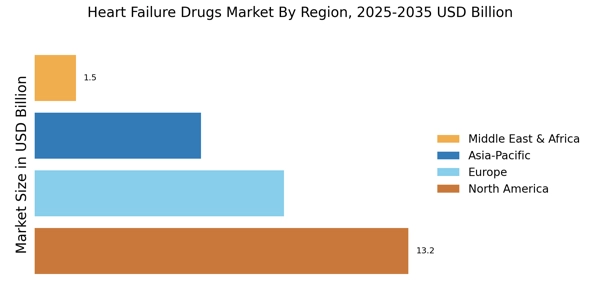
3. Market Segmentation
Market segmentation analysis is done according to type, application and geography.
i. Type:
This segmentation is done based on the type of drugs used for the treatment of heart failures such as ACE inhibitors, Angiotensin II antagonists, Beta-blockers, Diuretics and others.
ii. Application:
This segmentation is done on the basis of end-user applications of heart failure drugs such as cardiology centres, hospitals, and clinics.
iii. Geography:
This segmentation is done according to geographic regions such as Europe, North America, Latin America, Asia Pacific, and the Rest of the World.
4. Research Approach
The research methodology used for this report includes both primary and secondary data. Primary data is collected through interviews and surveys conducted with individuals working in the heart failure drugs market and industry participants. Secondary data is collected from periodicals, government publications, investor presentations, third-party databases, and published sources for the heart failure drugs market. The data is analyzed using Porter’s Five Forces Model, SWOT analysis and other quantitative and qualitative methods.
5. Market Estimation
For the market estimation process, the weighted average formula is used, to sum up, the individual market share of each segment and region. All the data points are confirmed by industry experts or industry participants.
6. Validation
The data points are validated by conducting multiple secondary research sources along with primary sources to ensure data accuracy.
7. Analysis
The data collected through primary and secondary sources is analyzed using Porter’s Five Forces Model, SWOT analysis and other quantitative and qualitative methods. Market players’ market strategies and techniques are also analyzed and studied to understand their potential impact on the heart failure drugs market.
8. Market Forecast
The final market forecast is conducted after analyzing and studying the data collected from primary and secondary sources. The market size and trends of the heart failure drugs market are estimated and forecasted taking into consideration the CAGR, total market revenue, segment revenue, and Y-O-Y growth over a five-year period.
9. Assumptions
The market size and growth of the heart failure drugs market are estimated and forecasted using various assumptions such as the CAGR, total market revenue, segment revenue and Y-O-Y growth over a five-year period.