

- Global Market Outlook
- In-depth analysis of global and regional trends
- Analyze and identify the major players in the market, their market share, key developments, etc.
- To understand the capability of the major players based on products offered, financials, and strategies.
- Identify disrupting products, companies, and trends.
- To identify opportunities in the market.
- Analyze the key challenges in the market.
- Analyze the regional penetration of players, products, and services in the market.
- Comparison of major players’ financial performance.
- Evaluate strategies adopted by major players.
- Recommendations
- Vigorous research methodologies for specific market.
- Knowledge partners across the globe
- Large network of partner consultants.
- Ever-increasing/ Escalating data base with quarterly monitoring of various markets
- Trusted by fortune 500 companies/startups/ universities/organizations
- Large database of 5000+ markets reports.
- Effective and prompt pre- and post-sales support.
eClinical Solution Market Size Snapshot
| Year | Value |
|---|---|
| 2024 | USD 8.9 Billion |
| 2035 | USD 20.4 Billion |
| CAGR (2025-2035) | 7.83 % |
Note – Market size depicts the revenue generated over the financial year
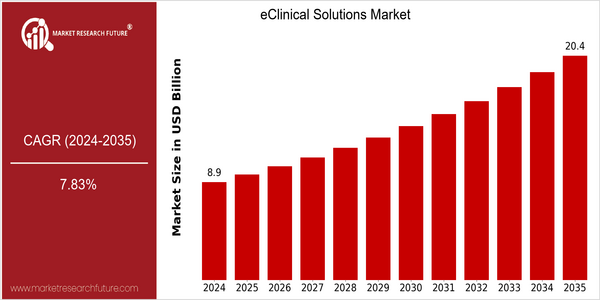
The eClinical market is poised for considerable growth, with a market size of $8.9 billion in 2024, which is projected to reach $20.4 billion by 2035. CAGR of 7.83% from 2025 to 2035. The rising demand for efficient clinical trial management and the growing use of digital technology in the health sector are the key growth drivers for this market. The increasing need for pharmaceutical and biotechnology companies to optimize their operations and data management capabilities is making eClinical solutions an indispensable tool in the clinical research landscape. Furthermore, technological advancements such as the integration of artificial intelligence and machine learning in clinical data management are further bolstering market growth. These innovations enable more accurate data analysis and patient recruitment strategies, thereby improving the overall efficiency of clinical trials. The leading players in the eClinical solutions market, such as Medidata, Oracle, and Veeva, are focusing on strengthening their market positions through strategic alliances and product launches. These collaborations, which aim to develop cloud-based solutions and increase data interoperability, are a clear indication of the industry’s commitment to utilizing technology to improve clinical outcomes.
Regional Deep Dive
E-clinical solutions market is experiencing a considerable growth across different regions. The increasing demand for efficient clinical trial management and the adoption of advanced technology have been driving the growth of the e-clinical solutions market. The North America e-clinical solutions market is characterized by a high concentration of key players, a strong healthcare system, and a high regulatory compliance. Europe is characterized by a diverse landscape and varying regulatory frameworks. The Asia-Pacific region is emerging rapidly owing to increasing investments in the healthcare and technological industries. Middle East and Africa are experiencing a steady growth, owing to the government initiatives to improve the healthcare system. Latin America is gradually adopting e-clinical solutions as a part of the digital transformation of the healthcare industry.
North America
- The recent interest in digital health has led to the development of eClinical solutions. Medidata and Oracle have been at the forefront in developing these solutions.
- In 2023 the National Institutes of Health launched a new initiative to integrate e-clinical solutions into clinical trials, to facilitate data collection and to increase patient involvement.
- The clinical trials landscape is changing. With the growing focus on patient-centricity and real-world evidence, organizations like the CTTI are advocating for more adaptive trial designs.
Europe
- The European Medicines Agency (EMEA) has published new guidelines for the use of electronic data in clinical trials. The new guidelines encourage the implementation of e-clinical solutions in all member states.
- In the e-clinical field, the new methods of artificial intelligence and machine learning are being integrated into the platforms, as companies such as Veeva and Covance have been doing in Europe.
- The regulatory environment varies from country to country, which makes it necessary to develop specific e-clinical solutions in order to comply with the requirements and to be efficient. This requires the collaboration between local and global players.
Asia-Pacific
- China and India are catching up fast on e-clinical solutions, thanks to the government’s drive to go digital and improve clinical trial efficiency.
- The advent of telemedical treatment and remote patient monitoring during the COVID-19 pandemic has increased the demand for e-clinical solutions, and companies such as Wuxi AppTec are expanding their operations in the region.
- eClinical solution companies and local biotech companies have been collaborating more and more, which is enhancing the region's ability to conduct complex clinical trials.
MEA
- The Gulf Cooperation Council (GCC) countries are investing heavily in health care. This has resulted in a greater interest in eClinical solutions, in order to facilitate clinical trials.
- Regions are beginning to establish a framework for digital health. The Saudi Food and Drug Authority, for example, has begun to promote the use of digital data in clinical research.
- Hence the emergence of local health institutions and international eClinical solution suppliers, which seek to improve clinical trial processes and the care of patients.
Latin America
- Brazil and Mexico are two countries where e-clinical solutions are in great demand. In both countries the need for more efficient clinical trials management is being met by the need for greater access to health care.
- The Pan American Health Organization (PAHO) supports the integration of digital health solutions into clinical research, which would increase data quality and patient engagement.
- eClinical companies, addressing the local challenges, are emerging and establishing themselves in the local markets. These companies offer a new source of innovation and competition.
Did You Know?
“About seventy percent of clinical trials fail to meet their recruitment targets.” — Clinical Trials Transformation Initiative (CTTI)
Segmental Market Size
The e-clinical market is growing rapidly, driven by an increasing need for efficient clinical trial management and data integration. The real-time data required for clinical trials, regulatory pressure to get new drugs to market faster, and the increasing adoption of electronic health records (EHRs) which allow seamless data sharing between different parties are the key growth drivers of the e-clinical market. In the e-clinical market, companies like Medidata and Oracle have emerged as leaders by providing comprehensive platforms to improve the efficiency and accuracy of clinical trials.
Currently the market is in the midst of a period of expansion, with a marked adoption in North America and Europe, where the regulatory framework is more favorable to the development of new clinical practices. The main applications are those of data capture, clinical trial management systems and patient engagement tools, which are essential for the success of clinical trials. A pandemic like the scourge of influenza accelerated the shift towards remote monitoring and decentralization of trials. The development of artificial intelligence and machine learning will determine the future of clinical data analysis. These tools are essential to optimize the design of trials and to improve the recruitment of patients.
Future Outlook
The e-clinical solutions market is expected to grow at a CAGR of 7.83% from 2024 to 2035. This growth will be driven by the increasing adoption of digital health technology, the increasing need for clinical trial management, and the increasing trend towards decentralization of clinical trials. The penetration of e-clinical solutions is expected to increase significantly, with a penetration rate of over 70% in clinical research organizations by 2035, compared to an estimated 40% in 2024.
IT has a key role to play in e-clinical, and with the advent of new technology, the collection of data, the participation of the patient and the conduct of the trial will be greatly improved. In addition, the integration of EHRs with e-clinical solutions will make data sharing easier and improve patient care. The emergence of new trends, such as the focus on patient-centricity and the importance of real-world evidence, will also change the market dynamics. The market will become a major component of the clinical trial system, and the clinical trial system will be able to improve the speed and quality of drug development.
Covered Aspects:| Report Attribute/Metric | Details |
|---|---|
| Market Size Value In 2022 | USD 7.28 Billion |
| Market Size Value In 2023 | USD 8.24 Billion |
| Growth Rate | 13.2% (2023-2032) |
eClinical Solution Market Highlights:
Leading companies partner with us for data-driven Insights
Kindly complete the form below to receive a free sample of this Report
Tailored for You
- Dedicated Research on any specifics segment or region.
- Focused Research on specific players in the market.
- Custom Report based only on your requirements.
- Flexibility to add or subtract any chapter in the study.
- Historic data from 2014 and forecasts outlook till 2040.
- Flexibility of providing data/insights in formats (PDF, PPT, Excel).
- Provide cross segmentation in applicable scenario/markets.









