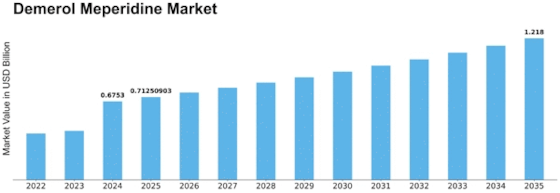
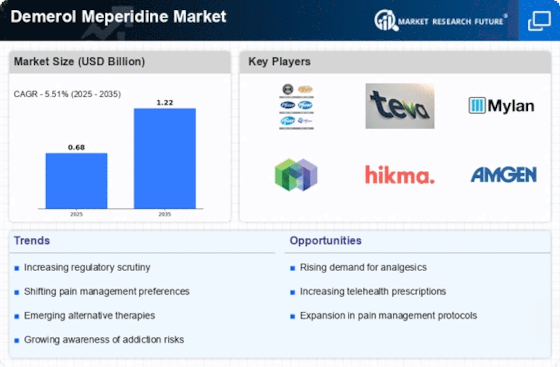
Demerol Meperidine Size
Demerol Meperidine Market Growth Projections and Opportunities
The Demerol (Meperidine) market is heavily influenced by broader trends in pain management. The prevalence of pain-related conditions and the medical community's approach to addressing pain play a significant role in shaping the demand for Demerol and other analgesic medications. Various opioid analgesics and other pain management products create the competitive environment in the Demerol market. Efficacy, pricing, and safety profile together with the likeness to other pain medications affect the market share. Age, gender, and specific medical conditions among the patients are also the market factors in the case of Demerol. Certain patients population like those who are in severe pain post-surgery or those with chronic pain conditions are among the patients that creates the demand for this opioid analgesic. The market for Demerol is influenced by the opioid epidemic as well as concerns about drug abuse. Demerol perception and utilization in the medical community are affected by the more strict controls, prescribing guidelines, and measures aimed at combating the misuse of opioids. The market of Demerol is awarded by the occurrence of several medical conditions related with severe pain including postoperative pain, cancer-related pain, or specific acute conditions. The occurrence of these conditions adds to the need for the efficient pain management drugs. The price of Demerol and its accessibility to patients as well as health systems are important market factors. Pricing policies as well as the presence of generic substitutes influence the market’s accessibility to various demographic groups. The prescribing behaviour of healthcare professionals including physicians and surgeons plays on the demand for Demerol. Changes in the prescription rules or the shift to alternative pain relief strategies can influence the market trends. The research and development activities in pain management and analgesic medications influence the market of Demerol. Development of new analgesics or improved safety profile formulations can cause changes in market preferences. The demand for Demerol is influenced by the public perception of opioids as well as patient education initiatives. Greater knowledge of the perils and advantages and other pain management techniques can influence the market via the choices and preferences of the patients. Global health indicators, such as the incidence of particular diseases or health conditions necessitating analgesics, also contribute to the global Demerol demand. Differences in healthcare infrastructure and drug availability also determine market dynamics.
Technological advancements in pain management, such as the development of non-opioid alternatives or innovative drug delivery systems, impact the Demerol market. Evolving technologies contribute to the diversification of pain management options.



















Leave a Comment