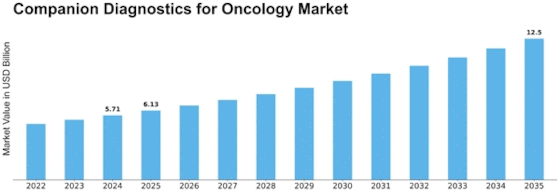
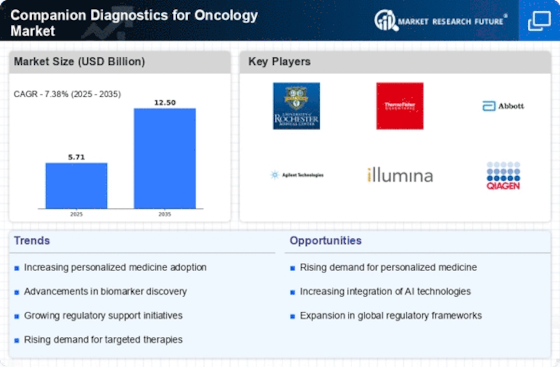
Companion Diagnostics Oncology Size
Companion Diagnostics Oncology Market Growth Projections and Opportunities
Companion diagnostic (CDx) tools have revolutionized the landscape of personalized medicine, particularly within the pharmaceutical industry. These innovative testing kits serve as integral components in the development and implementation of tailored therapeutic solutions. They operate by scrutinizing genetic material to furnish critical insights into the optimal utilization of corresponding treatment protocols. The marriage of CDx tests with therapeutic products has engendered a paradigm shift in healthcare, steering it towards a more individualized approach.
Amidst a backdrop of escalating cancer incidences, the role of companion diagnostics has attained paramount significance. Their ability to decipher and leverage genetic information has become indispensable in the oncology sphere. As the prevalence of various cancer types continues to surge, the need for precise, targeted therapies intensifies. Companion diagnostics, thus, emerge as pivotal tools in discerning the most effective treatment strategies, steering away from generalized approaches towards highly personalized interventions.
This surging demand for companion diagnostics in oncology stems from several factors. Primarily, the imperative to enhance therapeutic outcomes while mitigating adverse effects drives pharmaceutical companies and healthcare providers alike to embrace these sophisticated tools. By tailoring treatments based on genetic profiles, CDx tests hold the promise of optimizing therapeutic efficacy, thereby potentially minimizing unwarranted side effects and improving patient outcomes. This symbiotic relationship between diagnostics and therapeutics underscores the transformative potential of companion diagnostics in reshaping the healthcare landscape.
Furthermore, the surge in strategic initiatives has propelled the adoption and integration of companion diagnostics into clinical practices. Collaborations between diagnostic companies, pharmaceutical giants, and regulatory bodies have fostered an environment conducive to the advancement and utilization of these cutting-edge technologies. Such partnerships facilitate the development of robust diagnostic platforms, ensuring their regulatory compliance and seamless integration into healthcare systems worldwide.
Moreover, the burgeoning research endeavors aimed at elucidating the intricate interplay between genetics and disease pave the way for the continuous evolution of companion diagnostics. Advancements in genomic profiling techniques and bioinformatics augment the precision and scope of these diagnostic tools, enabling a more comprehensive understanding of diseases at a molecular level. Consequently, this burgeoning knowledge reservoir amplifies the efficacy and applicability of companion diagnostics, propelling their demand and utilization across diverse clinical domains.



















Leave a Comment