Cardiac Biomarkers Market Summary
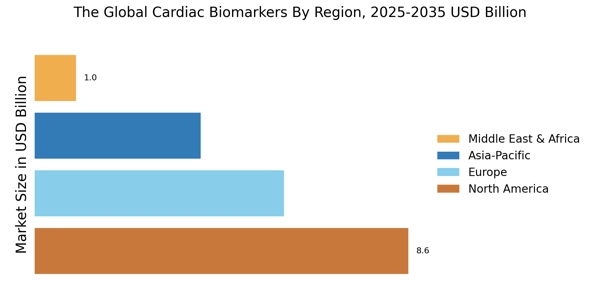
According to Market Research Future analysis, the Cardiac Biomarkers Market Size was valued at USD 19.2 Billion in 2024. The cardiac biomarkers industry is projected to grow from USD 21.51 Billion in 2025 to USD 66.93 Billion by 2035, registering a CAGR of 12.02% during the forecast period (2025–2035). North America led the market with over 44.79% share, generating around USD 8.6 billion in revenue.
The Cardiac Biomarkers Market is primarily driven by rising cardiovascular disease prevalence and increasing demand for early and accurate diagnosis, encouraging adoption of advanced biomarker testing solutions that improve clinical decision-making, enable timely interventions, and enhance overall patient outcomes globally.
According to World Health Organization, cardiovascular diseases cause approximately 17.9 million deaths annually, representing 32% of global deaths. This high burden positively drives demand for cardiac biomarkers, supporting early diagnosis, improved treatment outcomes, and expanding adoption of advanced diagnostic technologies worldwide.
Key Market Trends & Highlights
The Global Cardiac Biomarkers Market is poised for substantial growth driven by technological advancements and increasing healthcare demands.
- Technological advancements in diagnostics are enhancing the accuracy and efficiency of cardiac biomarker testing.
- North America holds 44.79% share, with United States contributing 82% of regional revenue in 2024.
- Point of care testing dominates with 58% share, driven by rapid diagnostics and increasing emergency cardiac care demand.
- Troponins segment leads with 42% share due to high accuracy in early myocardial infarction detection globally.
- Congestive heart failure segment holds 28% share, growing rapidly due to rising global cardiovascular disease prevalence.
Market Size & Forecast
| 2024 Market Size | 19.2 (USD Billion) |
| 2035 Market Size | 66.93 (USD Billion) |
| CAGR (2025 - 2035) | 12.02% |
Major Players
Companies such as Roche (CH), Abbott (US), Siemens Healthineers (DE), Thermo Fisher Scientific (US), Ortho Clinical Diagnostics (US), BioMérieux (FR), QuidelOrtho Corporation (US), Boehringer Ingelheim (DE), F. Hoffmann-La Roche AG (CH) are some of the major participants in the global market.