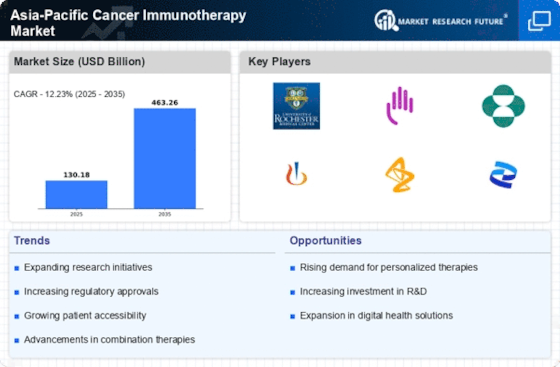
Market Analysis
In-depth Analysis of Asia Pacific Cancer Immunotherapy Market Industry Landscape
Cancer immunotherapy is benefiting from Asia Pacific's rising cancer diagnosis rate. The prevalence of senior people, lifestyle changes, and the environment are contributing to a rise in some cancers in the area. Immunotherapy for cancer is new in Asia-Pacific. Using the immune system to fight cancer is a growing industrial trend. It appears like a potential alternative to traditional cancer immunotherapy.
Immunotherapy has a lot of untapped potential, and Asia-Pacific countries are spending money to find new ways to use it. It is now possible to treat cancer better thanks to the work of the government and groups that are not with either of them.
Many of the companies in the Asia-Pacific area that make cancer vaccines work in the science and drug industries. They make and sell the most cutting edge immunotherapeutic drugs on the market right now, which makes them a lot of money.
People with cancer can get help at a lot of study sites and clinical studies in this area. Different groups such as drug companies, schools, and study groups are working together to find out more about immunotherapeutics and how they can be used on people. This is a project for a group.
Inside the Asia-Pacific area, there are a lot more patient support groups and events that teach people about health services. To help more people learn about cancer immunotherapy, these groups want to make it easy to get more information about it. They also want to promote early cancer diagnosis and treatment so that many patients can have better results.
A "checkpoint inhibitor" is a type of immunity drug that is becoming better known in the Asia-Pacific room. These medical treatments are meant to get rid of chemicals that weaken and hurt the immune system. When these things get into the body, they help the immune system find and kill cancer cells.
The market is hurt by the fact that healthcare systems in Asia and the Pacific can't talk to each other. Possible that rich countries spend more on health care than less developed countries because their processes are better developed. Anyhow, countries that aren't as well developed might have trouble with things like cost and access. Medical professionals, academic groups, and drug companies are talking to each other more these days. People can share tools and knowledge through these relationships. This will speed up the process of making immunotherapies for cancer in the long run.
What makes this possible is the price difference between biosimilars and real biologics. The market gets used to them every once in a while. Biologics are being used more and more as Asian and Pacific health care systems try to find the best mix of how well they work and how much they cost.
Each country in the Asia-Pacific area has its own rules about how to give and keep track of immunotherapeutic drugs. People from that country are the only ones who can follow these rules. So that new drugs can be used quickly and correctly, the government needs to make its processes clearer and more uniform.
There are changes in the drugs used to treat cancer in Asia and the Pacific because more and more people are getting personalized care. Biomarker study is becoming more popular because it helps doctors figure out what makes each patient special and pick the best drugs for them. The medicine works better this way, and it can be changed to fit the person.



















Leave a Comment