Rising Prevalence of Chronic Diseases
The North America Interventional Radiology Products Market is experiencing growth due to the increasing prevalence of chronic diseases such as cardiovascular disorders, cancer, and diabetes. According to the Centers for Disease Control and Prevention, chronic diseases account for approximately 70% of all deaths in the United States. This alarming statistic drives the demand for advanced interventional radiology procedures, which are often less invasive and more effective than traditional surgical methods. As healthcare providers seek to improve patient outcomes, the adoption of interventional radiology products is likely to rise. Furthermore, the aging population in North America, which is more susceptible to these diseases, further fuels the market. The combination of these factors suggests a robust growth trajectory for the North America Interventional Radiology Products Market.
Growing Preference for Outpatient Procedures
The North America Interventional Radiology Products Market is witnessing a shift towards outpatient procedures, driven by patient preferences and healthcare cost considerations. Patients increasingly favor minimally invasive procedures that allow for quicker recovery times and reduced hospital stays. According to a report from the American College of Radiology, outpatient interventional radiology procedures have seen a significant rise, with many patients opting for these alternatives to traditional surgeries. This trend not only enhances patient satisfaction but also reduces healthcare costs, making it an attractive option for providers. As more healthcare facilities adopt outpatient models, the demand for interventional radiology products is likely to grow. This shift towards outpatient care is indicative of a broader transformation in the North America Interventional Radiology Products Market, emphasizing efficiency and patient-centered care.
Regulatory Support for Innovative Treatments
Regulatory support for innovative treatments is a key driver of the North America Interventional Radiology Products Market. Agencies such as the Food and Drug Administration (FDA) are actively promoting the development and approval of new interventional radiology products that demonstrate safety and efficacy. The FDA's expedited review processes for breakthrough devices facilitate quicker access to advanced technologies for healthcare providers and patients. This regulatory environment encourages innovation and investment in the interventional radiology sector, as companies are more likely to pursue research and development of novel products. As regulatory frameworks continue to evolve in favor of innovation, the North America Interventional Radiology Products Market is poised for growth, with an increasing array of products entering the market to meet the demands of healthcare providers.
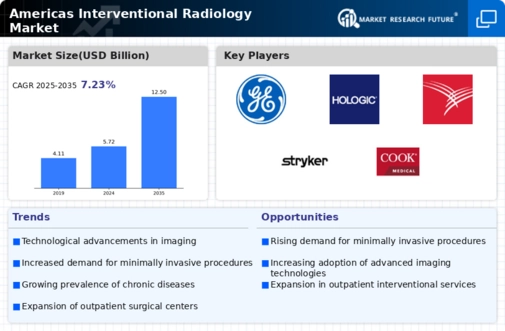
Technological Innovations in Imaging Techniques
Technological advancements in imaging techniques are significantly influencing the North America Interventional Radiology Products Market. Innovations such as 3D imaging, MRI-guided interventions, and advanced ultrasound technologies enhance the precision and effectiveness of interventional procedures. These technologies not only improve diagnostic accuracy but also facilitate real-time monitoring during procedures, thereby reducing complications and enhancing patient safety. The integration of artificial intelligence in imaging is also emerging, potentially streamlining workflows and improving decision-making processes for healthcare professionals. As hospitals and clinics invest in state-of-the-art imaging technologies, the demand for interventional radiology products is expected to increase. This trend indicates a promising future for the North America Interventional Radiology Products Market, as healthcare providers strive to adopt the latest innovations to improve patient care.
Increased Investment in Healthcare Infrastructure
The North America Interventional Radiology Products Market is benefiting from increased investment in healthcare infrastructure. Governments and private entities are allocating substantial funds to enhance healthcare facilities, particularly in the realm of advanced medical technologies. This investment is crucial for the development and expansion of interventional radiology services, as it enables hospitals to acquire the latest equipment and technologies necessary for effective patient care. For instance, the U.S. government has implemented various initiatives aimed at improving healthcare access and quality, which indirectly supports the growth of interventional radiology. As healthcare infrastructure continues to evolve, the demand for interventional radiology products is expected to rise, reflecting the industry's adaptability to changing healthcare landscapes. This trend underscores the potential for sustained growth within the North America Interventional Radiology Products Market.


















Leave a Comment